Chapter 6: Introduction to Neuro-ophthalmology
A third of the brain is devoted to the visual system, so neurology is an important topic! While the rest of ophthalmology involves image recognition (your ability to recognize corneal abrasions, disk cupping, and infections under the microscope), neuroophthalmology requires a more “cerebral” analysis. This makes learning the subject painful at first – trust me, it becomes more entertaining as you progress! For our purposes, I’m going to keep things simple and only cover topics that you should know as a medical student.
FUN FACT:
 Phrenology is the study of the morphology of the skull, and was developed by Franz Josef Gall (1758 – 1828). Gall felt there was a direct link between the shape of the skull and human character and intelligence. While complete bunk, Gall was one of the first to consider the brain the source of all mental activities.
Phrenology is the study of the morphology of the skull, and was developed by Franz Josef Gall (1758 – 1828). Gall felt there was a direct link between the shape of the skull and human character and intelligence. While complete bunk, Gall was one of the first to consider the brain the source of all mental activities.
Phrenology was very popular in America throughout the 1800’s and its influence can still be seen in our language. For example, people with “high brows” were considered more intelligent than those with “low brows.”
Diplopia:
A common complaint you’re going to be faced with is “double vision.” Patients frequently complain of doubling — and sometimes they actually mean it! Often, however, they just mean that their vision looks blurry. Technically, the phrase diplopia describes the symptom of seeing two different images of the same object, and that’s what we are going to discuss!
When faced with a diplopic patient, there is an important question you must immediately answer: before breaking out your arsenal of neuroophthalmic flags, prisms, and muscle lights, you must distinguish whether the diplopia is monocular or binocular. If the double-vision remains when you cover an eye then you have a monocular diplopia. You should breath a sigh of relief at this point – because monocular double vision isn’t a neurologic problem at all and your exam just got easier!
Monocular doubling is often caused by a refractive problem in the front part of the eye. There aren’t any mechanisms of monocular doubling that occur at the retina or further back in the neuro pathway. The most common cause of monocular diplopia is astigmatism, an abnormal curvature of the corneal surface. New onset astigmatism could occur from corneal deformation from an overlying lid lesion or after surgery with tight corneal stitches through the cornea. Other causes of monocular diplopia include cataract irregularities, lens displacement, or primary problems with corneal curvature such as keratoconus.
Binocular diplopia, on the other hand, occurs when the eyes do not move in synchrony with each other. This can occur from nerve lesions (a palsy of CN3, CN4, or CN6), extraocular muscle abnormalities (such as the muscle-fibrosis that occurs with Grave’s Disease), or derangements at the neuromuscular junction (myasthenia gravis). Let’s explore the cranial nerve palsies first.
Cranial Nerves and EOMs
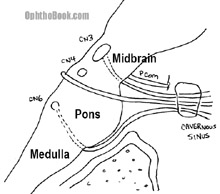 Three cranial nerves control the movements of the eyeballs. The relationships of these muscles can be quite complex as the eyeballs are neurologically “yoked” together and every muscle has multiple vectors of force, depending upon the direction that the eye is looking. Nerve palsies can be challenging to work out!
Three cranial nerves control the movements of the eyeballs. The relationships of these muscles can be quite complex as the eyeballs are neurologically “yoked” together and every muscle has multiple vectors of force, depending upon the direction that the eye is looking. Nerve palsies can be challenging to work out!
There are numerous causes for the individual nerve palsies, including microvascular disease, strokes, tumors, and aneurysms. If you’re like myself, you’re probably not up-to-speed on your neuroanatomy. I’ve drawn this cartoon picture of the brainstem for you to reference over the next few pages.
Third Nerve Palsy
Oculomotor nerve pasly is the easiest to detect because a complete 3rd nerve palsy looks dramatic. The majority of the extraocular muscles are innervated by CN3, so when knocked-out the eye deviates down and out because of the still functioning abducens and superior oblique muscles. In addition, the levator palpebrae (the main lid retractor) is innervated by CN3 and its paralysis gives you a severe eyelid ptosis. Finally, the parasympathetic pupil-constrictor fibers from the Edinger-Westphal nucleus travel within CN3, and their loss gives you a “blown pupil.”
 Most third nerve palsies are caused by ischemic events at the nerve secondary to hypertension or diabetes. The one thing you really need to worry about in these patients is a compressive aneurysm pushing on the nerve. These aneurysms occur at the junction of the posterior communicating artery and the internal carotid artery. Compressive lesions usually affect the parasympathetic nerve component: a blown pupil is a potential emergency. Whenever you have pupillary involvement, you need an MRI and angiography to rule out a dangerous aneurysm or tumor.
Most third nerve palsies are caused by ischemic events at the nerve secondary to hypertension or diabetes. The one thing you really need to worry about in these patients is a compressive aneurysm pushing on the nerve. These aneurysms occur at the junction of the posterior communicating artery and the internal carotid artery. Compressive lesions usually affect the parasympathetic nerve component: a blown pupil is a potential emergency. Whenever you have pupillary involvement, you need an MRI and angiography to rule out a dangerous aneurysm or tumor.
Pupil Involvement with CN3:
Oculomotor palsies often have pupillary involvement because the parasympathetic nerves innervating the iris travel with the third nerve. Pupillary involvement is a crucial diagnostic sign — compressive lesions tend to involve the pupil, while vascular lesions actually spare it! This picture isn’t drawn to scale, but graphically demonstrates what I’m talking about.
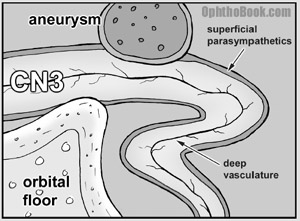
As you can see, the parasympathetic nerves course along the surface of the oculomotor nerve making them susceptible to compressive lesions from the outside such as an aneurysm from the posterior communicating artery, boney structures, or the uncal portion of the temporal lobe. Ischemic lesions (caused by HTN and diabetes) occur deeper within the oculomotor nerve and thus spare the superficial parasympathetic fibers.
If you have a patient with CN3 loss and pupillary involvement you should order an MRI and an angiogram to look for the compressive site. If there isn’t pupillary involvement, they are probably suffering from a vaso-occlusive problem, so you should check their glucose and blood pressure.
Abducens (VI):
 The abducens nerve controls the lateral rectus muscle. Loss of CN6 renders the eye unable to abduct (turn out). Patients will go cross-eyed, so to compensate they may turn their head to avoid double vision.
The abducens nerve controls the lateral rectus muscle. Loss of CN6 renders the eye unable to abduct (turn out). Patients will go cross-eyed, so to compensate they may turn their head to avoid double vision.
If you look back in that drawing of the brainstem, you’ll see that the abducens nerve is located further down the brainstem, “all by its lonesome” down in the pons. The nerve root exits the brainstem even further down at the ponto-medulary junction and has to run up the floor of the skull to get to the cavernous sinus and into the orbit. Where the nerve enters the cavernous sinus, it makes an abrupt 90-degree bend. Something about this abrupt turn makes the 6th nerve susceptible to high intracranial pressure. Patients with high ICP from pseudotumor cerebri commonly have their 6th nerve(s) knocked out – an abducens palsy is actually incorporated into the Dandy criteria for diagnosing PTC.
FUN FACT:
Crocodiles shed tears, but this isn’t a sign of grief. These secretions help shed salt-water from the eye. Thus, the term “crocodile tears” is used to describe false tears. In ophthalmology, we use the term to describe aberrant regeneration after 7th nerve injury – nerves that normally control salivation are routed to the lacrimal gland. This makes you “cry” when you see food. This can be treated by injecting botox into the lacrimal gland.
Aberrant regeneration occurs with other cranial nerve palsies as well — the most commonly seen is after a 3rd nerve palsy. As the oculomotor nerves grow back to their muscles they can get mixed up. For example, a patient could look medially (activating their medial rectus) and their eyelid can shoot up (inappropriate co-contraction of the levator palpebrae).
You only get this kind of synkinesis with trauma or mass lesions that disrupt the nerve sheath. Microvascular events occur deeper in the nerve and don’t cause aberrant regeneration. If you find aberrant degeneration in a patient you previously assumed was from diabetes, you better get imaging to rule out something more dangerous.
Trochlear Nerve (IV):
 The trochlear nerve (CN4) innervates the superior oblique muscle. Trochlear paralysis is the hardest cranial nerve palsy to diagnose and many ophthalmologists and neurologists will miss these! These patients have an upward deviation of the affected eye and a “cyclotorsion” twisting of the eye that makes them tilt their head away from the lesion. Don’t try to memorize these deviations: in a few paragraphs I’ll cover the anatomy of the superior oblique muscle which will make it easier to conceptualize these findings.
The trochlear nerve (CN4) innervates the superior oblique muscle. Trochlear paralysis is the hardest cranial nerve palsy to diagnose and many ophthalmologists and neurologists will miss these! These patients have an upward deviation of the affected eye and a “cyclotorsion” twisting of the eye that makes them tilt their head away from the lesion. Don’t try to memorize these deviations: in a few paragraphs I’ll cover the anatomy of the superior oblique muscle which will make it easier to conceptualize these findings.
A trochlear nerve lesion is caused by either trauma, an ischemic event, or can be congenitally present with later decompensation. The fourth cranial nerve is the skinniest nerve and runs the longest distance inside the cranial vault. This long passage makes it more susceptible to injury if the brain sloshes around and bounces against the tentorium. The fourth nerve is also susceptible to being pulled from the root where it exits from the back of the brainstem. The long course also makes it susceptible to neoplasm. If we break down trochlear palsy by cause:
1/3rd Trauma
1/3rd Congenital
1/3rd Ischemic (diabetic)
1/3rd Tumor
That’s a lot of thirds, I know. That’s because reports differ depending upon what age-group you look at: certainly more 4th palsies occur in elderly males from trauma and more congenital palsies are found in the pediatric population. Ask about history of closed-head injuries and check old photographs for head-tilt – this would indicate an old/congenital palsy that has recently decompensated.
Troclear Muscle Action:
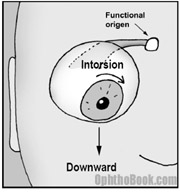 The superior oblique muscle runs from the back of the orbit, forward through a trochlear “pully” located next to the upper nasal bridge, before inserting at the back of the eye. This pulley system completely changes the direction of force of the superior oblique … you can think of the trochlear pulley as the “functional origin” of this muscle.
The superior oblique muscle runs from the back of the orbit, forward through a trochlear “pully” located next to the upper nasal bridge, before inserting at the back of the eye. This pulley system completely changes the direction of force of the superior oblique … you can think of the trochlear pulley as the “functional origin” of this muscle.
As you can see in this picture, the superior oblique muscle inserts onto the back of the eyeball and then yanks the eye down. There is also an intorsional component that rotates the 12-o’clock corneal limbus towards the nose. This explains the head-tilt these patients develop.
A deeper peek at the superior oblique:
To simulate the action of the superior oblique, you can pretend that your head is a large eyeball. Throw an arm up and wrap it around the back of your head. Your elbow becomes the trochlear pully … if you pull your arm, you’re whole head should twist. The direction of head movement, either up-down or rotational, will depend upon which direction you’re looking when you start pulling.
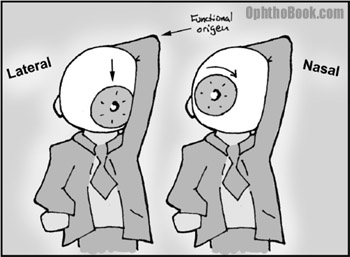
The same action occurs in the eyeball such that your patient will see vertical diplopia when looking medially toward the nose (such as when reading a book) and will see more rotational doubling when looking to the side. Think about that one for a minute!
Summary of the EOMs:
Beyond the information above, there isn’t much to localizing a cranial nerve lesion. Just think about the anatomy: if a single nerve is affected, then you know the problem is somewhere along that nerve’s tract. If all three nerves are knocked out, then the lesion is probably near the cavernous sinus where the three nerves are bunched together.
The number one reason that any of the cranial nerve gets knocked out is from a vasculitic event, usually from diabetes. Many of these isolated cranial nerve palsies don’t need imaging – such as an isolated 6th nerve palsy in an elderly diabetic. However, you don’t want to miss an aneurysm or mass lesion, so no one will fault you for over-imaging. Here are the high-yield facts you should know:
- CN3: The eyes are “down and out” with a droopy eyelid. Think of an aneurym if the pupil is blown.
- CN4: Patient tilts their head away from the lesion. Think of trauma or a congenital head-tilt that has decompensated with age.
- CN6: The patient looks “cross-eyed.” Consider increased intracranial pressure.
Myasthenia Gravis:
Myasthenia gravis is a rare autoimmune disease in which the body develops autoimmune antibodies to the nicotinic acetylcholine receptors located at the neuromuscular junction of striated muscle. This leads to fatigable muscles and often involves the eye, causing diplopia and ptosis.
MG patients develop autoantibodies that actually bind to the receptor and block the receptor binding sites, and eventually destroy the receptor entirely, leaving patients with decreased numbers of Ach receptors. Once the number of receptors drops below 30% normal, then the patient becomes symptomatic, with easily fatigability. Interestingly, only striated muscle is affected, as both smooth and cardiac muscle appear to have different antigenicity and are unaffected with this disease. The bulbar muscles, however, are quite susceptible, and the majority of patients with MG have ocular complaints – the ophthalmologist is often the first doctor to diagnose this disorder.
 The diplopia and ptosis is usually worse on prolonged upgaze: you can test this by having your patient look at your raised finger to see who tires out first. More definitive diagnosis can be made via the Tensilon test where you give edrophonium chloride (an anticholineresterase) and look for an improvement in symptoms as their Ach levels build up. We don’t actually do this test in our office because of the difficulty of starting IV lines and potential toxic reactions such as sweating, salivation, bronchospasm, and bradycardia. More commonly we’ll perform a rest-test or ice-test where you have the patient hold an icepack over their closed eyes and then remove it and look for improvement. Neurology can also perform EMG studies and other tests to help you with this diagnosis.
The diplopia and ptosis is usually worse on prolonged upgaze: you can test this by having your patient look at your raised finger to see who tires out first. More definitive diagnosis can be made via the Tensilon test where you give edrophonium chloride (an anticholineresterase) and look for an improvement in symptoms as their Ach levels build up. We don’t actually do this test in our office because of the difficulty of starting IV lines and potential toxic reactions such as sweating, salivation, bronchospasm, and bradycardia. More commonly we’ll perform a rest-test or ice-test where you have the patient hold an icepack over their closed eyes and then remove it and look for improvement. Neurology can also perform EMG studies and other tests to help you with this diagnosis.
Systemically, these patients can have problems with mastication, talking, drinking, and swallowing. Aspiration pneumonia and respiratory failure from inability to clear secretions is the big killer with this disease. Remember, if your patient has MG, work them up for a thymoma and check their thyroid levels.
Neuritis and Neuropathies of the Optic Nerve
Personally, I always found this topic confusing because the terms “optic neuritis” and “optic neuropathy” sound very similar. After all, what’s the difference between an “itis” and an “opathy?” There’s really only three main optic-nerve entities that I think you need to be aware of, and each has a slightly different presentation and mechanism:
1. ON (Optic Neuritis)
An “inflammation” of the nerve, often demyelinating. The cardinal signs in these patients are decreased vision (especially color vision), pain with eye-movement, enhancement of the optic nerve on MRI, and potential association with multiple sclerosis. Occurs in younger patients.
2. ION (Ischemic Optic Neuropathy)
… sometimes called NAION (non-arteritic ischemic optic neuropathy)
This is a localized ischemic event at the junction of the optic nerve as it enters the back of the eyeball. This portion of the optic nerve has no elastic “give” and a small vascular insult here can lead to swelling and vision loss. The hemispheric vascular supply to the optic nerve head usually generates an altitudinal visual defect. This entity usually occurs in middle-age in those with a predisposed crowded optic disks (the so-called “disk at risk”).
3. GCA (Giant cell arteritis) i.e. temporal arteritis
Temporal arteritis occurs as a result of a vasculitis within the medium and small-sized arteries around the head. The vasculitis can lead to a sudden occlusion of the blood supply to the eye leading to sudden and permanent vision loss This happens in older patients, usually over 70 years of age.
Let’s explore each of these entities in more detail …
Multiple Sclerosis and Optic Neuritis:
Multiple sclerosis is a demyelinating disease of the CNS that is classically described as “lesions occurring at different times and different places.” It occurs most commonly in young white women from northern climates. If the lesion hits the optic nerve, then we call this finding optic neuritis. About 90% of patients with multiple sclerosis will develop optic neuritis at some point, and conversely, patients with “optic neuritis” sometimes progress to develop multiple sclerosis. Think about that one for a moment!
Signs and symptoms of optic neuritis include:
– Sudden vision loss (central scotoma is classic)
– Decreased contrast and color sensitivity
– Pain with eye movement
– Optic nerve head edema
– Afferent pupillary defect
A patient with optic neuritis needs an MRI of the brain and orbits to look for enhancing lesions. The more demyelinating lesions found on imaging, the higher the chance of later developing multiple sclerosis. Patients with optic neuritis are treated with IV steroids, which will speed recovery, but won’t ultimately affect the outcome of the disease. WARNING: You treat with IV steroids only, as oral steroids may actually increase the reoccurrence of MS! If enhancing lesions are found in the brain, then you can get neurology involved to discuss possible treatment with interferons like Avonex to decrease progression.
FUN FACT
People blink, on average, once every 5-6 seconds.
Women blink almost twice as often as men.
Temporal Arteritis:
Temporal arteritis (also known as giant cell arteritis) is an important syndrome to keep in the back of your head. While not terribly common, you might save a patient from complete blindness or death if you treat them appropriately.
Temporal arteritis is an inflammation that affects the medium-sized blood vessels. This disease process is similar to polymyalgia rheumatica except that the vasculitis affects the arteries supplying the head, face, and eyes. If the blood supply to the eye is affected, then patients can have catastrophic vision loss. These patients are almost always older (over 60 and more commonly over 80 years of age) and present with sudden, painless vision loss. Other preceding systemic complaints (these are pathognomonic) include:
– Scalp tenderness and headache
– Jaw claudication
– Polymyalgias of the arms and shoulders
– Fevers, night sweats, weight loss
If you suspect GCA you need to order an ESR and CRP as these are sensitive markers for inflammation. Normal ESR is approximately half the patient’s age (i.e., an 80-year-old man can have ESR up to 40). Unfortunately, these labs aren’t very specific and more definitive diagnosis is made via a temporal artery biopsy (dissect out the artery at the temple and send it to pathology). On pathology you’ll find disruption of the internal elastic lamina and occasionally giant cells (the presence of these cells isn’t actually necessary for the diagnosis).
You treat temporal arteritis with steroids to decrease inflammation. While steroids won’t regain lost vision, they will decrease potential vision loss in the remaining eye, which can be affected within days. Unfortunately, steroids also decrease the diagnostic yield on your biopsy. This places you in a dilemma: do you hold off steroids until after the biopsy, or start steroids and potentially mess up your biopsy results? Well, the answer is that you start the steroids immediately to keep the other eye from being affected. You don’t want to blind your patient – the biopsy can be delayed for up to two weeks and still be ok, despite the steroids.
The Pupil
The pupil is controlled by a steady balance between the parasympathetic (which constricts the pupil) and the sympathetic input (which dilates the pupil). I always remember this with the mneumonic:
“If a grizzly bear attacks me in the dark woods, my sympathetic fight-or-flight reflex dilates my eyes so I can see better as I run away.”
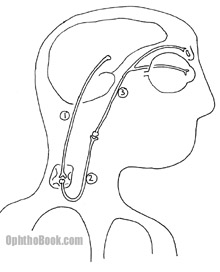
Horner Syndrome
Horners occurs when the sympathetic pathway gets knocked out. Without sympathetic input to the pupillary dilator muscles, the pupil constricts and stays small. Sympathetic loss also creates a mild ptosis from decreased Muller’s muscle action in the eyelid and scalp anhydrosis (decreased sweating) on the affected side. As you can see in the picture below, the sympathetic chain is long, complex, and can be damaged at many levels. To localize the lesion we use a series of eyedrops:
Cocaine Test
 The first test we perform is the cocaine test – just to decide if this patient REALLY has a Horners pupil or not. Cocaine stimulates pupillary sympathetics by decreasing norepinephrine uptake at the synaptic cleft. If a patient (a Horner’s patient) has no sympathetic tone inside the eye, then cocaine won’t have any norepinephrine to build up and thus has no effect on that eye. However, the good eye will dilate like the dickens! It’s not always easy to obtain cocaine in private practice, but you should be able to get some in most hospitals as ENT people use it to control nose bleeding during surgery.
The first test we perform is the cocaine test – just to decide if this patient REALLY has a Horners pupil or not. Cocaine stimulates pupillary sympathetics by decreasing norepinephrine uptake at the synaptic cleft. If a patient (a Horner’s patient) has no sympathetic tone inside the eye, then cocaine won’t have any norepinephrine to build up and thus has no effect on that eye. However, the good eye will dilate like the dickens! It’s not always easy to obtain cocaine in private practice, but you should be able to get some in most hospitals as ENT people use it to control nose bleeding during surgery.
Paradrine Test
Now that we know there’s a sympathetic palsy, we need to localize the lesion. There are three neurons in the pathway from the brain… just like the muscles motor-innervation throughout the rest of the body. Unlike a leg muscle, however, we can’t check the pupillary reflex by hitting it with a hammer. We can stimulate that final 3rd order nerve by pharmacologically hammering it with hydroxyamphetamine. This drug forces the end-nerve to fire-away at the pupil. If that pupil still won’t dilate, then you know the final “lower motor neuron” is dead. If the pupil DOES dilate, then you must have a “higher order” nerve that’s out – lots of bad things can occur along this upper-pathway (carotid dissections, pancoast tumors, etc.) so procede to imaging.
This pharmacological testing is a convoluted topic, and you’ll find it difficult to really remember these drops until you see your first Horner’s patient. One catch phrase you should remember, though: if a patient complains of a painful Horners think of a carotid dissection and move quickly to rule out this diagnosis.
Adie’s Tonic Pupil
An Adie’s pupil is the opposite of a Horner’s – the parasympathetic (constrictor) pathway gets knocked out on its way to the iris sphinter muscles. On exam, the eye is dilated and doesn’t constrict to light (we’re blocking the parasympathetic pathway from the Edinger-Westphal nucleus). The pupil will constrict with near vision- but very slowly. That’s why we call it a “tonic pupil,” it’s tonically slow.
Fortunately for us, the parasympathetic pathway is much shorter than the convoluted sympathetic pathway, so potential causes are more benign. The parasympathetic plexus sits right behind the eye and can be damaged after an otherwise benign viral infection.
PIMP QUESTIONS
1. You have a patient with diplopia. His left eye is turned down and out and his lid is ptotic on that side. What nerve do you suspect and what should you check next?
This sounds like a CN3 palsy, and you should check his pupillary reflex. Pupillary involvement suggests the lesion is from a compressive source such as an aneurysm.
2. Why do diabetic patients with oculomotor paralysis have “sparing of their pupil?”
The pupil is typically spared with iscemic third nerve palsies caused by vascular problems. This is because the parasympathetic pupillary fibers run along the surface of the nerve, making them susceptible to aneurysm/tumor compression but resistant to deeper infarction.
3. This 32 year old overweight woman complains of several months of headaches, nausea, and now double vision. What cranial nerve lesion do you see in this drawing. What other findings might you expect on fundus exam and what other tests might you get?

This looks like a bilateral abducens palsy as the patient can’t move either eye laterally. While the majority of abducens palsies occur secondary to ischemic events in diabetics and hypertensives, this etiology seems unlikely in a young patient with bilateral involvement. Her symptoms sound suspicious for pseudotumor (obese, headaches). You should look for papilledema of the optic nerve, get imaging, and possibly send her to neurology for a lumbar puncture with opening pressure.
4. A patient is sent to your neurology clinic with a complaint of double vision. Other than trace cataract changes, the exam seems remarkably normal with good extraocular muscle movement. On covering the left eye with your hand, the doubling remains in the right eye. What do you think is causing this diplopia?
The first question you must answer with any case of diplopia is whether the doubling is monocular or binocular. This patient has a monocular diplopia. After grumbling to yourself about this inappropriate neurology referral, you should look for refractive problems in the tear film, cornea, lens, etc..
5. A patient complains of intermittent double vision that seems to be worse in the evenings. On exam you find a confusing diplopia that doesn’t seem to map out to any particular nerve palsy. What else is on your differential as a cause, and what tests might you perform in the office?
Myasthenia gravis and thyroid orbitopathy are both great masqueraders that cause diplopia. Graves patients often have lid retraction and reduced upgaze from inferior rectus muscle restriction. The double vision in myasthenia patients can look like an isolated nerve palsy, a mixture of nerve involvement, or may not fall into any specific nerve combination – a changing palsy is more indicative of a process like MG. You can check for fatiguable ptosis by prolonged upgaze (hold your arm up and see who gets tired first). In addition, you can perform a cold-pack rest test or even a Tensilon test.
6. You are giving a tensilon test to a suspected myasthenia gravis patient and he collapses. What do you do?
Your patient may have a reaction to the anticholinersterase such as bradycardia or asystole. You should have a crash-cart handy and administer atropine. Hopefully, this scenario never happens to you. In this day and age, few ophthalmologists perform the tensilon test, reserving this for neurology (who more often perform EMG studies).
7. A patient with diplopia is finally diagnosed with myasthenia gravis after a positive ice-pack test and a positive acetylcholine receptor antibody test. What else should you work up this patient for.
You should check for a thymoma, which is highly associated with MG. Also, check their thyroid level as 20% of myasthenia patients also have Grave’s disease.
8. A 26 year old woman presents with decreased vision in her left eye that has gotten progressively worse over the past week. The eye seems to ache and the vision worsens with exercise. On exam she is found to have 20/200 vision, trace APD, and markedly decreased color vision in the affected eye. The optic nerve is mildly swollen on that side. What does this patient most likely have?
This patients age, color vision, and progression are all classic symptoms of optic neuritis. She also describes the classic Uthoff phenomenon of worsening symptoms with body-temperature (exercise or shower). Many of these patients describe minor pain with eye-movement: the optic nerve is inflamed and any tugging on the nerve with eye movement is going to irritate it.
9. A patient develops optic neuritis. Should you treat with steroids? Would you start with IV or oral steroids? Will the MRI findings of numerous demyelinating lesions change your management? Do you tell the patient that she will develop MS?
The ONTT study has shown that steroids can speed recovery from optic neuritis, but have little effect on long-term visual outcome. Surprisingly, the study also showed that oral prednisone may actually increase reoccurrence of optic neuritis. Therefore, you give IV Solu-Medrol and don’t give oral prednisone!
The presence of optic neuritis does not necessarily mean the patient will develop multiple sclerosis, especially in the setting of a negative MRI. The patient’s long-term risk for developing multiple sclerosis depends upon the number of CNS lesions found on presentation. If there are no CNS lesions, then the future risk is only about 15%. This jumps up to 50% or more with 3+ lesions. In these higher-risk patients, you should get neurology involved to discuss more aggressive treatment with Avonex.
10. An 84-year-old man was out golfing with his buddies and developed sudden vision loss in his right eye. He has no past ocular history, no medical problems. No complaints of flashes or floaters, just that things “look dimmer” in his right eye. What other questions should you ask about his symptoms?
There are many questions you should ask … but with any elderly person with vision loss, be sure to ask about the symptoms of temporal arteritis. Specifically, scalp tenderness, jaw claudication, and polymyalgias (muscle aches in the shoulders and arms). This sounds like a central retinal artery occlusion, and in a patient this old you need to rule out life- and vision-threatening causes like GCA.
11. The patient admits to “not feeling good” and “it hurts my head to brush my hair on the right side” for the past week but denies all other symptoms. Should you order any labs? Start any medications?
If you have any suspicion for GCA, you pretty much have to order a ESR and CRP. Start oral prednisone (about 1mg/kg/day) immediately and set up for temporal artery biopsy within a week or so. Steroids won’t help much with his lost vision in these cases, but decreases the risk to the other eye, which can be affected within hours to days.
12. A young man complains of complete vision loss (no light perception) in one eye, however, he has no afferent pupil defect. Is this possible? How might you check whether this patient is “faking it?”
Assuming the rest of the eye exam is normal (i.e. the eye isn’t filled with blood or other media opacity) this patient should have an afferent pupil defect if he can’t see light. There are many tests to check for malingering: you can try eliciting a reflexive blink by moving your fingers near the eye. One of my favorite techniques is to hold a mirror in front of the eye. A seeing eye will fixate on an object in the mirror. Gentle rocking movements of the mirror will result in a synchronous ocular movement as the eye unconsciously tracks the object in the mirror.
The best ophthalmology book!, the videos are amazing!!
Thanks, this is phenomenal.
alhamdulilah. can’t thank you enough.
Excellent! Superb! The Author transferred the more complicated subject to much more simpler one. ‘Piece of cake’. I recommed this site to all medical students and practising Ophthalmologists and Optometrists.
Nice. Ophthal made easy 🙂
thanks alot
wonderful!thanks alot.
nice book
please tell me, if a patient with a pupil that doesnt dilate as much as the other, with the difference being at least 2mm, and the cocaine test dilates the eye, what else could it be?
This book,is terrific.Understanding of it,is easy.Good work
The content is simplified easy to study and understand highly recomended and the videos are great
Sir,
On the action of superior oblique in your video lecture, I get this doubt. When the right eye is abducted the right superior oblique intorts the right eye. When the right eye is in primary position(gazing straight) the right superior oblique will again intort the right eye. Would you say that comparing the intortion in two scenarios, the intortion in a abducted eye will be more than the intortion in a primary gazed eye?
I was wondering what could cause my grandmother in her 80’s to experience double vision, crosseyes, massive headaches…it was mistakeningly diagnosed as temporal arteritis but on further examination of the piece of artery they extracted, was that it was healthy and was not temporal arteritis…The Doctors are baffled this is her second time in 10 years with the same symptoms on the same area…Any thoughts I just wanna see her get better quick
this was really helpful. i’m still struggling to figure out my intermittent vertical double vision – binocular. i’m 50 yr old female. it occurs in the a.m. and last approx 90 minutes. it’s been 2+ years.
i only wish i had visited this site earlier, hats off to all the people behind it
Thank you so much. This is a wonderful resource.
Ran into a tree doing 80km was in a coma for 5days have no memory of accident have problems seeing and have a big problem eating I can’t have anything cold as my lips feels like they are frost bitten and inside my mouth it is very painful when I chew instead of taste I feel like electric impulses. When I drink it is painful I can only drink warm tea, water at room temp starts this electric charge in my mouth.I can not walk easy, on uneven ground it is about impossible it feels like the ground is moving around me. I feel like I am in a different reality. I have seen all sorts of Specialists and one says 4th nerve Palsy!No clear answers. Do you have any suggestions? Thank you
I have had keracatonus for30+ years and in the past few years being tested for Ms do you think there is a link there. I am vittualy blind now
My left eye is so much smaller than the right eye not only that but my pupils dont move unless i move my head from side to side i dont even blink much. What could be the problem what should i do ?
Im a 57 year old woman with 4th nerve palsy which came on suddenly 1 year ago. May be a congenital condition with decompensation due to stress, as some earlier photos do reveal a head tilt. I wear prism glasses which help–but fatigue and any alcohol send me into double vision hell. Am considering surgery–and wonder on chances of success?
57 year old woman with vertical double vision in both eyes. Worse with fatigue. no ptosis. Letters on page align with tilting head back. Many articles talk about a side head tilt but not the tilt straight back. What does this suggest?
This is a great resource. Thank you.
Question for you – Diplopia, Paragraph 2, Sentence 2 – Is it supposed to read the way it does? I am not tracking. Please review at your convenience.
thanks ^^
great book
This is the most amazing website I have ever come across. Why didn’t I found this during my first year of university?!
My husband has undiagnosed double vision in one eye only. He gets in intermittently for periods of weeks or months and then it goes for up to 8 or so months. He is struggling to see the right person, being sent back every time to see opthalmists who do the same tests, each time concluding that there is nothing wrong with the eye. Can you help in any way by letting me know who would be a good specialist to contact?
Hello,
I have a scenerio for you if you would be so kind as to share your input on the subject. I am a 58 year old female with an approximate 3 month history of severe headache, noted mostly on awakening in the morning. Headaches are so severe I refer to them as “brain thumpers” as I can also hear and feel my heart pounding in my ears when they occur. I have a history of some mild to moderate hypertention and have had an ablation for SVT a few years ago. Approximately a month ago I noticed that my right pupil was not centered in my eye when I was looking straight forward. I noticed it while looking in the mirror when doing my hair. I thought it was my imagination at first and then my son in law noticed it one day and said something to my daughter about it. The right pupil is pulling to the right side (or outer aspect) of my eye and sometimes appears to be pulled slightly upward. I sometimes have mild pain on movement of my eye and often feel like I have to blink a few times to focus my vision. I have had a few episodes of double vision as well. In general I feel terrible. I am very fatigued and of course realize I am more than likely suffering some anxiety over my eye and headaches. My neck feels stiff pretty much all day everyday. I am on B/P meds as follows metoprolol 50 q day, Losartan 50 q day which was just increased to 100 mg yesterday due to continued elevation of my blood pressure which has been ranging from 160’s to mid 170’s systolic and from high 80’s to low 100’s diastolic. I have had a CT scan to r/o brain tumor and/or aneurysm. I am scheduled for an MRI, MRA, and MRV next Wednesday and scheduled to see an optomologist in the coming days- though the appt has not been set yet. What is your impression, given my symptoms, as to what the issue could be? Signifcant family history includes mutiple incidence of aortic aneurysms and aortic dissection. 36 year old brother deceased from aortic dissection, two sisters who survived aortic dissection-(both when they were 49 y.o.) one of which has dissected 4 times, an Aunt deceaseed from ruptured aneurysm, 2 cousins deceased from aortic dissection/ aneurysm, one of which expired post repair when his repair “let go”. My materal grandmother is said to have died from cerebral hemorrhage at he age of 49 though records are difficult to retrieve to substantiate.
Thank you,
Becky Brown
I’m in my final year at medical school. I think opthalmology is definitely my weakest point and I is not something I have ever had a huge interest in. But I decided to overcome that and I am sooo grateful I found this book! It’s absolutely fantastic!
I have been diagnosed with sixth nerve palsy, it has got slightly better, how long will this take to be perfect.? Does ice, botox etc help or do you have to have the muscle stitched as the nerve is not sending the right signals
75 year old male, cataract surgery Jan. 20, 2013, left eye, Alcon Toric lens. In recovery room, noticed that vision was excellent except tilted down from right to left, approx. 5 deg. Advice was to get right eye done, see if brain corrects problem. Right eye done Mar. 13. After 3 months now, no improvement. Neuro opthomologist confirms surgery properly done, says wait 6 months. Says may be trauma to 4th optic nerve during surgery.
Any advice or other observations?
Thanks
I am a 60 year old male with crossed eyes caused by a head injury. I was struck from behind by a car while riding a bicycle to work. I went up over the hood of the car and my head hit the windshield. I was unconsisous for some time. I have recovered in most ways. But I am still cross eyed in my left eye. It looks to the right down and leans to the right. I can align both eyes left to right, up and down. But as soon as I do the left eye’ s vision fades, and move back to the right and down again. When I first start trying to align them I felt a sharp pain in my left eye. But fortunately that has stop.
When I walk in the mornings the street appears to lean to the right in my left eye. My right eye is okay. Maybe leaning to the left just a little. But I can use it for getting around, riding a bike and mowing grass.
Since the accident in October of 2005 I have tried many eye exercises for improving my eyes. But since I am crossed eyed, I also have recurring lazy eye. Even though I can build up my vision from time to time. My lazy will pull it back down. I have always been able to straighten them up left to right and up and down, but never able to correct the left eyes’ s slant to the right.
One more thing, I have retina pigmentosa. At the time of the accident. I had a 20 percent field of vision. I know it did not cause the crossed eyes, but it sure is not helping. Since that I have found eye exercises that help it a whole lot and I do mean a lot. I have a wider field of vision, and my night time vision has improved wonderfully. But I am still crossed eyed. It is really been a problem for me. Is there any eye exercises for the leaning problem in the left eye? Any help would go a long ways, and be appreciated even more.
Thank You for Your time and Consideration
Steve Burton
I am not sure my condition is covered here.
My problem is I see the same object with both eyes,for ex. a square drawn on a white sheet of paper. My right eye always sees the square as it should. My left eye sees the square and it is tilted to the right. If I raise my head i can get left eye square to cover the right eye square. But if I lower my head the tilted square goes down and to the right.
Hi,
I see that the last comment was posted in November 2013 so I hope that I am not late for the much needed help.
I am 47 years old, and have suddenly and recently (past three weeks) experienced ptosis in my left eye lid and restricted movement in the right eye causing diplopia in the low to full right gaze. I also feel lightheaded and walking and focusing is quite hard without closing one eye. My eyes are healthy ophthalmology wise (according to the latest exam) apart from astigmatism.
My blood tests are very good including thyroid. My MRI shows nothing. STIMREP and SFEMG tests show only slight intermittent delay in my facial muscles around the eye (something is 27% and it should be below 23%) but the rest are very normal. the neurologists suggested Ocular Myasthenia and put me on Mestinon and for last seven days I see no improvement. My chest Xray is normal. Tried acupuncture, no improvement. Diplopia is constant and does not improve with rest.
I am (or rather was) healthy with good diet and regular exercise.
I would very much appreciate if you would be able to suggest any other tests I should do to diagnose the problem and eventually treat it.
Thanks and kind regards
DS
It is possible to have the author information for a MLA citation from this web page ?
Here is a link to author information.
made matters very INTERESTING and is useful not only to students but to seniors also.ingenious illustrations.
all the best
Macular pucker in one eye can also cause double vision. That’s a defect in the retina, not a nerve problem. I have it. Membrane peel surgery six months ago has provided a return of sharp vision but objects still appear displaced, distorted and larger in one eye than in the other.
great explanation
Thanks for making neuro -ophthalmology so simplified and enjoyable to grasp.