Chapter 3: Introduction to Glaucoma
Glaucoma is a disease where the optic nerve dies. We are not sure why or how this happens (there are many mechanical, vascular, and biochemical theories) but high intraocular pressure certainly seems to be associated, if not entirely the cause, of optic nerve death. Glaucoma is one of the leading causes of preventable blindness in the U.S., and patients with acute glaucoma can develop irreversible vision loss within a few hours, so it is important that you understand how this disease works and recognize it in your patients.
The Aqueous Pathway
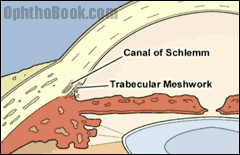 Before continuing we should review the pathway of aqueous humor flow. Aqueous is the fluid that fills the front part of the eye, and it is important for maintaining the shape of the eye and providing nourishment for the avascular lens and cornea. Aqueous humor is first produced by the ciliary body within the posterior chamber. After filling the posterior chamber, aqueous moves forward around the lens and flows through the pupil into the anterior chamber. As the anterior chamber fills, the aqueous spreads outwards into the angle formed by the iris and cornea. Within this irido-corneal angle the aqueous exits the eye by filtering through the trabecular meshwork into the Canal of Schlemm, where it returns back into the blood circulation. The pressure within the eye is maintained by this steady state of aqueous production and egress, and it is an imbalance in this equilibrium that causes the increase pressure associated with glaucoma.
Before continuing we should review the pathway of aqueous humor flow. Aqueous is the fluid that fills the front part of the eye, and it is important for maintaining the shape of the eye and providing nourishment for the avascular lens and cornea. Aqueous humor is first produced by the ciliary body within the posterior chamber. After filling the posterior chamber, aqueous moves forward around the lens and flows through the pupil into the anterior chamber. As the anterior chamber fills, the aqueous spreads outwards into the angle formed by the iris and cornea. Within this irido-corneal angle the aqueous exits the eye by filtering through the trabecular meshwork into the Canal of Schlemm, where it returns back into the blood circulation. The pressure within the eye is maintained by this steady state of aqueous production and egress, and it is an imbalance in this equilibrium that causes the increase pressure associated with glaucoma.
Open vs. Closed-Angle Glaucoma
There are two categories of glaucoma and they have very different mechanisms. Open-angle glaucoma is the most common type in our country. It occurs from decreased aqueous drainage caused by an unidentified dysfunction or microscopic clogging of the trabecular meshwork. This leads to chronically elevated eye pressure, and over many years, gradual vision loss.
This differs from closed-angle glaucoma, also called “acute glaucoma,” which occurs when the angle between the cornea and iris closes abruptly. With this closure, aqueous fluid can’t access the drainage pathway entirely, causing ocular pressure to increase rapidly. This is an ophthalmological emergency and patients can lose all vision in their eye within hours.
Let’s examine each of these types of glaucoma in more detail.
Open-Angle Glaucoma
 The majority of glaucoma patients (about 80% ) have chronic open angle glaucoma. Most patients are over the age of 40. This condition is more common in African Americans, and has a strong familial inheritance. The major risk factors are family history, age, race, high eye pressure, and large vertical nerve cupping. More recently, thin-corneas have been found to be a major risk factor, though this mechanism is not well understood.
The majority of glaucoma patients (about 80% ) have chronic open angle glaucoma. Most patients are over the age of 40. This condition is more common in African Americans, and has a strong familial inheritance. The major risk factors are family history, age, race, high eye pressure, and large vertical nerve cupping. More recently, thin-corneas have been found to be a major risk factor, though this mechanism is not well understood.
The underlying mechanism for open-angle glaucoma involves degeneration of the trabecular meshwork filter, usually by unknown causes, that leads to aqueous backup and chronically elevated eye pressure. With prolonged high pressure, the ganglion nerves in the retina (the same nerves that form the optic nerve) atrophy. The exact mechanism for this nerve damage is poorly understood and proposed mechanisms include stretching, vascular compromise, and glutamate transmitter pathways. As the ganglion nerves are progressively destroyed, vision is gradually lost.
 Open-angle glaucoma has the reputation of being the “sneaky thief of sight” because the visual loss occurs so slowly that many patients don’t realize they have the disease until it is far advanced.
Open-angle glaucoma has the reputation of being the “sneaky thief of sight” because the visual loss occurs so slowly that many patients don’t realize they have the disease until it is far advanced.
Because the disease is otherwise asymptomatic, detecting open-angle glaucoma requires early pressure screening. Free screening clinics also use different types of automated visual-field testing to detect subtle peripheral vision loss.
Presentation
Open-angle glaucoma patients usually present with three exam findings: elevated eye pressures, optic disk changes, and repeatable visual field loss patterns.
1. Pressure: The most accurate way to measure eye pressure is with the Goldman applanation tonometer. This is a device mounted on the slit-lamp that measures the force required to flatten a fixed area of the cornea. Normal pressures range from 10 to 22 mm Hg, while glaucoma patients can measure over 22 mm Hg. Keep in mind that eye pressure can fluctuate throughout the day (typically highest in the morning) so the pressure should be checked with each visit and the time of measurement should be noted. Also, some glaucomatous eyes have a “normal” pressure. In other words, a “good pressure” doesn’t rule out glaucoma, nor does a high pressure necessarily indicate glaucoma.
You can also measure pressure with a device called a “Tono-Pen.” This expensive little device is handy for bed-bound patients and down in the emergency room, though it’s inaccurate in the wrong hands!
Corneal Thickness can affect your pressure measurement:
When we measure the pressure in the eye, we are actually measuring how much resistance we get when pressing on the cornea. This is analogous to kicking a car-tire with your foot or pressing your hand against a bicycle tire to estimate how much air pressure is inside. We do the same thing with the Goldman applanation tonometer mounted on the slit-lamp – we measure how much force it takes to flatten a 3mm diameter area of corneal surface.
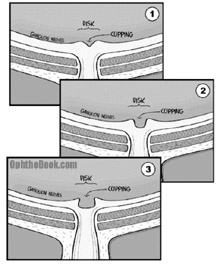 The pressure measurements on the Goldman were calibrated using an average corneal thickness of approximately 540μm. However, some patients have very thin or thick corneas. I like to describe these as “thin bicycle” or “thick truck-tire” corneas. When you press on a thick cornea (a truck-tire cornea) the pressure will seem higher than it really is! This makes sense … if you kick a flat truck-tire, it will still hurt your foot because that rubber is so darn thick. The opposite is true for thin corneas – they feel squishy no matter how much air pressure.
The pressure measurements on the Goldman were calibrated using an average corneal thickness of approximately 540μm. However, some patients have very thin or thick corneas. I like to describe these as “thin bicycle” or “thick truck-tire” corneas. When you press on a thick cornea (a truck-tire cornea) the pressure will seem higher than it really is! This makes sense … if you kick a flat truck-tire, it will still hurt your foot because that rubber is so darn thick. The opposite is true for thin corneas – they feel squishy no matter how much air pressure.
This thickness variability is important in glaucoma clinic so we can calibrate the accuracy of our pressure readings. This is why we always check corneal thickness with an ultrasonic pachymeter on the first visit.
2. Fundus Exam: The optic disk looks striking in advanced glaucoma. In normal patients, the optic disk has a physiological indentation or “cup” that is less than one-third the disk diameter. With glaucoma, the ganglion nerve layer slowly dies away, and, as fewer ganglion nerves course through the optic disk, the amount of cupping increases. A cup to disk ratio greater than 0.5 or an asymmetry between the eyes suggests ganglion atrophy caused by glaucoma.
The ISNT Rule
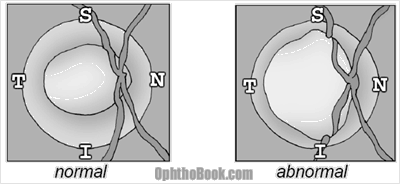
When you look at your patient’s optic nerve, you’ll notice that the cup doesn’t sit directly in the middle of the disk. The cup is slightly off-center – this makes sense as the optic nerve enters the back of the eyeball at an angle.
The space between the inner cup and surrounding disk is called the neural rim and is comprised of the actual retinal ganglion nerves. In a normal eye, this rim follows the ISNT rule of decreasing thickness … meaning that the Inferior rim is thickest while the Temporal rim is the skinniest.
With glaucoma, you often see vertical thinning and notching of the inferior and superior rims. This deviation from the ISNT rule is another clue that glaucoma might be killing off the nerve. You can also see undermining of the rim (like in the right drawing) where the blood vessels dive out of view under the rim edge.
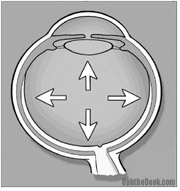
3. Visual Loss: The vision loss from chronic glaucoma occurs in characteristic patterns that can be followed by automated perimetry (machines that map out the peripheral vision). The central vision is typically spared – in fact, late stage patients may have 20/20 central vision, but be otherwise legally blind because of peripheral blindness.
FUN FACT!
The giant squid has the largest eyeball in the world. This enormous deep-sea predator can weigh up to 2.5 tons and 55 feet in length. Its eye can be a more than a foot in diameter! Wow!
Treatment:
Since IOP is the only risk factor we can treat, the primary treatment of glaucoma focuses on decreasing eye pressure to less than 20 mm Hg or even lower depending upon the severity of disease. Treatment may be either medical or surgical.
Medical Treatment
Topical beta-blockers are the traditional therapy for these patients and have been around for decades. Beta-blockers work by decreasing aqueous humor production at the ciliary body. Unfortunately, systemic side effects can occur from nasal absorption, making it especially important to ask your patients about history of asthma, COPD, and cardiac problems.
These days, many physicians are using newer drugs like topical CAIs, alpha-agonists, and prostaglandin analogues for first-line therapy, as they have fewer systemic side effects.
Prostaglandin analogues like latanoprost (Xalatan™) are the newest of these glaucoma drugs, and they are very popular as a first-line agent. They work by increasing aqueous humor outflow. They do have some side effects, though. They can make eyelashes grow longer (many patients actually like this), and in a few patients may darken the iris color, turning green and blue eyes brown.
Surgical Treatment for Chronic Glaucoma
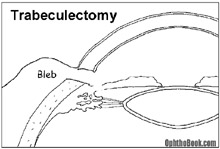 If eyedrops aren’t working, there are several surgical techniques available to relieve eye pressure. One common surgery is the trabeculectomy, where an alternate drainage pathway is surgically created. A small hole is cut through the superior limbus, creating a drainage tract from the anterior chamber to a space under the conjunctiva. This can be very effective in decreasing pressure, but if the patient is a rapid healer the shunt can scar down and close, so anti-metabolites like mitomycin-C are often applied to the site. If this surgery doesn’t work, a plastic tube-shunt can be inserted into the anterior chamber that drains to a plate fixed under the conjunctiva further back.
If eyedrops aren’t working, there are several surgical techniques available to relieve eye pressure. One common surgery is the trabeculectomy, where an alternate drainage pathway is surgically created. A small hole is cut through the superior limbus, creating a drainage tract from the anterior chamber to a space under the conjunctiva. This can be very effective in decreasing pressure, but if the patient is a rapid healer the shunt can scar down and close, so anti-metabolites like mitomycin-C are often applied to the site. If this surgery doesn’t work, a plastic tube-shunt can be inserted into the anterior chamber that drains to a plate fixed under the conjunctiva further back.
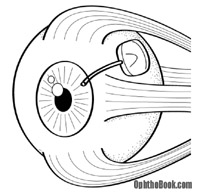
Several laser procedures can also help. Argon laser trabeculoplasty (ALT) can be used to burn portions of the trabecular meshwork itself. The resulting scarring opens up the meshwork and increases outflow. A laser can also be used to burn the ciliary body to decrease aqueous production at its source.
Acute Glaucoma
Acute glaucoma is a medical emergency. The most common mechanism is pupillary block. This occurs when the lens plasters up against the back of the iris, blocking aqueous flow through the pupil. This resistance produces a pressure gradient (this is a good buzz word to memorize) across the iris that forces the iris and lens to move anteriorly. When the iris moves forward, the irido-corneal angle closes, blocking the trabecular meshwork. Without an exit pathway, aqueous fluid builds up, eye pressure increases rapidly, and the retina is damaged from stretching and decreased blood supply.
The outflow angle can close for many reasons, and people with naturally shallow anterior chambers such as hyperopes (far-sighted people with small eyes) and Asians are predisposed to developing angle closure. One inciting condition that is typical in acute glaucoma is pupil dilation — many patients describe onset of their symptoms occurring while in the dark or during stressful situations. When the iris dilates, the iris muscle gets thicker and the irido-corneal angle becomes smaller, making it more likely to spontaneously close. Along those lines, medications that dilate the eye, such as over-the-counter antihistamines and cold medications, also predispose angle closure.
Presentation
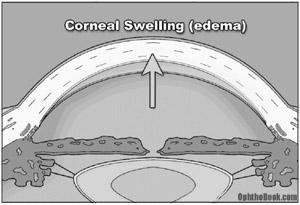
These patients will present with an extremely red and painful eye, often complaining of nausea and vomiting. On exam, you’ll find their pupil sluggish and mid-dilated. Pressures in the affected eye can be very high, often 60 mm Hg or higher. The eye will feel rock hard, and you can actually palpate the difference between the eyes with your fingers. One classic sign that patients often describe is seeing halos around lights. This occurs because the cornea swells as water is pushed under high pressure through the endothelium into the corneal stroma. This corneal swelling also makes it hard for you to see into the eye, further complicating diagnosis and treatment.
Acute Glaucoma Exam Techniques:
Ophthalmologic examination for acute glaucoma involves measuring the eye pressure, accessing the anterior chamber angle, and a fundus exam.
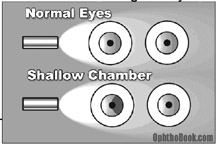 One trick to determine whether an angle is shallow is to shine a simple penlight across the eyes. If the iris is pushed forward, it will cast a shadow. Additionally, an ophthalmologist can visualize the angle directly through gonioscopy. Here’s how it works:
One trick to determine whether an angle is shallow is to shine a simple penlight across the eyes. If the iris is pushed forward, it will cast a shadow. Additionally, an ophthalmologist can visualize the angle directly through gonioscopy. Here’s how it works:
Gonioscopy:
Normally, the inside angle cannot be seen with a microscope because the cornea-air interface creates “total internal reflection.” However, we can use a goniolens, which is a special glass lens with mirrors on its sides, to look directly at the angle. When the glass lens is placed directly onto the cornea, the cornea-air interface reflection is broken and light from the angle can escape and be seen through the mirrors.
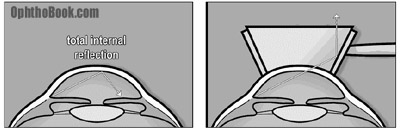
We can see the concept of “total internal reflection” in nature. For example, if you’ve ever gone snorkeling in water, you may notice that the water’s surface above you looks like a mirror. The flying fish uses this phenomenon to escape from predators. When attacked, the flying fish leaps from the water and glides above the surface so the shark can’t see them – effectively disappearing. This is also how fiber optic cables work, with light bouncing off the walls of the cable.
Acute Glaucoma Treatment
In cases of acute glaucoma, you want to decrease the pressure in the eye as quickly as possible. A “kitchen sink” approach is often used, throwing many treatments on at once. You can decrease aqueous production using a topical beta-blocker like Timolol and a carbonic anhydrase inhibitor like Diamox. Also, osmotic agents such as oral glycerin or IV mannitol (even ethanol, in a bind) can be given systemically to draw fluid out of the eye and back into the bloodstream. Finally, a miotic such as pilocarpine may be helpful in certain cases to constrict the pupil and thus open up the outflow angle. You can also use topical glycerin to transiently dehydrate/clear the cornea to aid with examination.
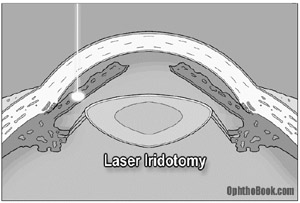 Ultimately, these patients need surgical treatment to avoid recurrence of their angle closure. A high intensity laser can burn a hole through the iris and create a communication between the posterior and anterior chambers, relieving the pressure gradient (buzzword!!) across the iris, and allowing it to move back into a normal position. This opens up the trabecular meshwork and allows aqueous fluid to flow freely out of the eye. This procedure is typically performed on both eyes because these patients are predisposed to having attacks in the other eye as well.
Ultimately, these patients need surgical treatment to avoid recurrence of their angle closure. A high intensity laser can burn a hole through the iris and create a communication between the posterior and anterior chambers, relieving the pressure gradient (buzzword!!) across the iris, and allowing it to move back into a normal position. This opens up the trabecular meshwork and allows aqueous fluid to flow freely out of the eye. This procedure is typically performed on both eyes because these patients are predisposed to having attacks in the other eye as well.
Other types of glaucoma
1. Neovascular Glaucoma:
This can occur in diabetic patients or those with a retinal vein occlusion. VEGF production from areas of ischemic retina can float forward through the pupil and promote neovascularization of the iris. In the early stages, a fibrous membrane forms on the iris-cornea angle that blocks outflow and forms an open-angle glaucoma. At later stages of neovascularization, the new vessels actually pull the iris forward and cause a closed angle glaucoma that is essentially irreversible. You treat this by lasering the peripheral retina to decrease the angiogenic VEGF production and decrease the rate of neovascularization. Neovascular glaucoma is very hard to treat and most of these patients end up needing a surgical intervention like a tube-shunt.
2. Pigment Dispersion Syndrome (PDS):
Occurs when the pigmented back-surface of the iris rubs against the radial zonules supporting the lens. Little flecks of pigment are shed into the aqueous and end up clogging the trabecular meshwork drain. This process usually occurs in young white males with myopic eyes. They suffer from attacks of high pressure after exercise when pigment gets rubbed off the iris. You can see this pigment in the trabecular meshwork on gonioscopy, and even find trans-illumination defects of the iris at the slit-lamp.
Some of the pigment will stick to the inner-corneal surface, and because of convection currents in the aqueous, form a vertical line of pigment on the inner corneal surface called a Krukenberg spindle.
3. Pseudoexfoliation Syndrome (PXF)
In this systemic condition, basement membrane-like material is deposited throughout the body. This material adheres to the anterior lens capsule, creating a rough surface. As the overlying iris dilates and contracts with daily activity, pigment is rubbed off and clogs the trabecular drain. These patients also suffer from zonular instability, making cataract operations difficult.
Summary
That is glaucoma in a nutshell. Chronic, open-angle glaucoma is very common (in this country) and leads to gradual visual loss, while acute closed-angle glaucoma is infrequent but an emergency that needs urgent treatment to avoid blindness.
PIMP QUESTIONS
1. What is glaucoma? What actually causes damage to the neurons and optic nerve with glaucoma?
Nobody is sure exactly “what glaucoma is” but at its most basic, glaucoma is gradual death of the optic nerve. If an attending asks you this question, say “knowbody knows” or “death of the optic nerve.” If you say “high pressure” you’ll be laughed at (glaucoma specialists are odd ducks). The optic nerve damage arises from pressure, stretching, sheer forces, vascular compromise, or some kind of hormone regulator – we’re not sure of the mechanism.
2. What is the flow-pathway for aqueous fluid? Where is it made, and where does it leave the eye?
Aqueous is first produced by the ciliary body. It then flows forward through the pupil into the anterior chamber. Finally, aqueous drains through the trabecular meshwork and back into the venous system via the canal of Schlemm.
3. What’s the difference between open-angle and closed-angle glaucoma? How about chronic versus acute glaucoma?
Open angle is a common, chronic condition where aqueous drainage is impaired. Closed-angle glaucoma is caused by acute closure of the irido-corneal angle leading to blockage of ALL aqueous drainage – an ophthalmologic emergency that can quickly lead to blindness.
4. What are the risk factors for developing primary open-angle glaucoma?
This is an important list and since I glossed over them in the chapter, here they are again:
High intraocular pressure (obviously)
Age
Family history
Race (African American and Hispanics)
Suspicious optic nerve appearance (large vertical cupping)
Thin central corneal thickness (** remember this one!)
There are other possible risk factors, but I’d focus on those above. These risk factors explain why we always ask our patients about familial history and why we check pachymetry (corneal thickness by ultrasound) on the first glaucoma visit.
5. What do we measure to monitor and follow progression in glaucoma patients?
We generally check three things: pressure, disk changes by 3D photograph, and visual fields. Good stereo slides are difficult to obtain, so many use other imaging modalities like HRT or OCT.
6. What does corneal thickness have to do with glaucoma (as far as risk for developing glaucoma)?
The OHTS trial showed that people with thin corneas are at higher risk for glaucoma, independent of other risk factors. We’re not sure why, but it’s believed that people with thin corneas are anatomically predisposed to optic nerve damage. We measure corneal thickness with a small ultrasound probe (this is called pachymetry) with all new glaucoma patients.
7. What’s a normal eye pressure? Does a patient with pressure of 14 have glaucoma?
About 10 to 22. While glaucoma is classically associated with high pressure, there is a significant minority of patients with glaucoma with normal pressure. Also, pressure fluctuates throughout the day and we typically write down the time in the notes. Some studies have noted higher rates of glaucoma in people with large diurnal shifts in eye-pressure. This observation could be compared to diabetic suspects with large diurnal shifts in random-glucose measurements.
8. A glaucoma suspect is found on first visit to have a pressure of 19. Her corneal thickness, however, measures only 450μm. Do you think her actual eye pressure is HIGHER or LOWER than 19?
Definitely higher. This patient has thin “bicycle-tire corneas” that “feel soft” when measured by the Goldman applanation. This woman’s actual pressure is probably well over 22, increasing her risk for glaucomatous damage progression.
9. What kind of vision loss occurs with glaucoma?
Typically loss of eyesight occurs in the periphery first where the loss is less noticeable. Scotomas (visual field loss areas) in glaucoma tend to follow certain patterns that start in the mid-periphery. Many patients don’t notice visual symptoms until the disease is far progressed. Generally, the central vision is spared until very late stages of glaucoma.
10. Why can’t you see the trabecular meshwork with the slit-lamp microscope?
Because the trabecular drain is behind the limbus (around the corner) and we can’t see this area directly because of “total internal reflection” at the cornea-air interface. Gonioscopy allows direct visualization of the trabecular meshwork by interrupting the cornea-air interface with a glass lens.
11. What mechanisms do the glaucoma drops use to decrease pressure?
Drops either decrease the amount of aqueous produced at the ciliary body or increase the aqueous outflow from the eye (generally via the uveal-scleral pathway or by direct trabecular meshwork outflow).
12. What retina findings do you see with glaucoma?
You see increased cupping of the optic disk, usually in a vertical pattern that goes against the ISNT rule. You can sometimes see hemorrhages at the disk and undermining of the blood vessels as they exit the disk.
13. How can diabetes cause acute glaucoma?
Retinal ischemia can produce VEFG. As this molecule floats forward it can cause neovascularization of the iris, forming vascular membranes that cover the trabecular meshwork and clog the drainage angle. This leads to a severe neovascular glaucoma that is hard to manage.
14. You have a patient who appears to have a shallow anterior chambers and occludable angles. Would you use pilocarpine?
In most cases, yes. Pilocarpine will constrict the pupils — by flattening the iris you potentially open up the drainage angle next to the trabecular meshwork. Pilocarpine will also decrease pressure in the eye by affecting aqueous production and egress. You probably wouldn’t use it long term in patients with occludable angles though, as pilo has a lot of side effects such as headache and blurry vision. Ultimately, anyone with occludable angles needs a laser peripheral iridotomy to equalize the pressure between anterior and posterior chambers.
thank you very much for this useful & wonderful
videos&chapters.
simple&cleare but needs more details on medical treatment
many many thanks ..really useful..
i like the idea of OPHTHOBOOK an i found it interesting and easy to understand. But apart from its goodness, its not complete. like you see in topic of glaucoma, there is no description about the visual feild defects occuring in different types of glaucoma. It must includ other topics like scleral diseases and many others.
in last i just want to thanks the OPHTHOBOOK as it has helped me alot!!!
Editor’s Note: Thanks for commenting. The purpose of this book is not to be an all inclusive textbook of ophthalmology, but to present these introductory topics in a simple and digestable format. Part of the challenge for me was to keep the chapters short – I had a very hard time keeping the book under 165 pages, and sometimes wish I’d made it shorter. I think this brevity is what makes this book useful and approachable. Some topics, such as the appearance of the glaucomatous visual fields that you mention, are just not high-yield topics for the neophite (though I do discuss these things in the videos). Also, other topics such as the scleral diseases (which are always highly covered in traditional texts) are not very important to the new student, either – I haven’t seen a case of scleritis or episcleritis in my clinic for at least six months. Any student going into ophthalmology must read larger (and more dauting texts) for this detailed information. Thanks for the constructive critisism!
Simply excellent…
simple but descriptive pictures for creating clearer concepts…
ZABARDAST (highest/best)
Another helpfull video 🙂
Any chance of another video or document outlining the glaucoma drugs in more deatail. I get soo confused trying to learn them all and make a real mess of trying to answer Qs about them in exams.
Thank you for taking the time to teach us
Many thank for the writer but need to explain some term clearly
weird text
On question 13, I believe that you meant to put VEGF rather than VEFG for the answer. Just wanted to let you know in case there is another publication. I love your book!!!
hey i was just wondering.. what the cause of the dilated pupil in glaucoma???
its realy vvvvvvvvvvv good no dought,,,,,
awesomeeeeeeee…………..
what is normotensive glaucoma?
i feel it is very informative and complete, i would like to get animation of glaucoma mechaism and other animated slide i am a part of glaucoma awarness screening programe .
Excellent book.it is a service for trainae doctors as well as service to humanity indireclottly.lovely/thanks a
Excellent book.it is a service for trainae doctors as well as service to humanity indirectly.lovely/thanks a lot
Please help. I am alarmed at the light that suddenly appears outside the rim of the lower eye. It looks like the eyelid itself with light in it. Sometimes my eyes are closed and I can still see this. It is now occurring with the other eye. I could be watching TV and reading a book and this occurs. Is it glaucoma. My family have glaucoma
Thank you
THANKS DOCTOR..
I LOVE IT..
CARRY ON..
many thanks for your wanderfull site, its a good learning site for medical students and those who wants to specialise in ophthalmology
Thank you very much for these important notes
Hello! I just got some questions about glaucoma..
When you say that glaucoma risk factors include family history, it is also important to screen for patients without any symptoms and with a positive family history for glaucoma.
But who exactly do you have to screen and when is it necessary to screen? eg. Girl, 22 years old and without any eye symptoms. Father is diagnosed with primary open angle glaucoma.. Necessary to already screen the girl at this age?
IOP is normal between 10 and 21 mmHg, but is it possible to have an IOP of 0 mmHg? If so, would it cause damage? If so, is it possible to ‘overtreat’ patients with glaucoma by use of any medical treatment?
Thanks alot!!
PS: your website is great!
Fantastic resource – we are doing optho right now and the lecturers tend to leap right into the small details and present them fairly dryly. Having this has helped greatly by providing a good foundation from which I can plug in the other smaller details I will need to memorize for the exam and then promptly forget… I’ve informed my classmates of this site and they have all been very impressed with it as well.
Thanks again,
Jamus
2nd year Canadian med student.
this is really a piece of art in presenting the glaucoma case thank u so much
I have a rare condition known as congenital ectropion uveae, which has created a problem for me of acute angle closure attacks. I would love to hear your understanding of the condition and how it affects the people who are plagued with it.
Thanks.
it would be even better if information regarding changes in visual field due to open angle glaucoma would have been given…
Thank you for your information. I recently had an exam where my IOP is 17, (appears to be normal range), discs are noted as flat, I have no family history of glaucoma and don’t fit the description of a glaucoma patient, yet my doc says he’s worried about it and wants me to come back for more tests. I’m skeptical and don’t like unnecessary testing.
Nice work
Keep it up…
First, I think your sight is spot on. Great job! Below is a short explanation of my experience. I hoe this is helpful to any current or future doctors. There is NO history of glaucoma of any kind in my family.
Topamax is BAD news. I went blind in 11 days on 14.5 mg (I cut the 25 mg pill in half) of Topamax. It caused acute angle closure glaucoma, with glaucoma flecks too. The sad thing is I was hospitalized twice and NO ONE looked at the side effects of the medication. Both times I told them it felt like an ice pick was being poked in my eye and that my vision was blurred they did nothing about it. Then after I went blind, the doctors (not ophthalmologist) said I went blind due to stress and continued to give me Topamax for 5 more days before they had and ophthalmologist see me. Who within hours figured out what had happened.
It is now over 8 years later and I am still blind. I share this with you so that others will be sure to check the side effects of the meds people are on. I was the 25th person the FDA said went blind as a direct result of Topamax.
Just like you said the pressure had to come down. I wouldn’t wish that pain on anyone. Laser surgery was done immediately after 5 different types of drops (that felt like acid) didn’t work to lower my eye pressure. Both my irises remain stuck dilated open, so when my eye pressure went down it caused all light to come in. Now and I guess you can say I am blinded by the light. Please remember to look at the uncommon side effects of meds that a person is on. I having never seen my grandson who just turned 5 years old, nor my youngest daughter since her third birthday she is now almost 12 years old. It has been very difficult. I pray your information gets to all those who need it. If I can help in any way please let me know.
Blessing to you always.
very informative site really
Thank you for this informative site!
it is liked the coolest sit ever i luv it =DDD
Excellent site! very useful when it comes to may eye subjects, takes you to the point of information. Keep it up!
simply superb…..
Thank you very much for this wonderful ophthalmology textbook and I like the sense of humor of the author. But I think there is a typo that the measuring unit of corneal thickness should be ‘μm’ rather than ‘nm’. Thanks again!
Thank you so much for this wonderful ophthalmology textbook and I like the sense of humor of the author. But I think there is a typo that the measuring unit of the corneal thickness should be ‘μm’ rather than ‘nm’. I love this book, too. Thanks again.
Thanks a billion. Because of these lectures I’m starting to take interest in Ophtholmology. You’re the best!
Thank you for that great job you simply did it .
wishing the best for you.
Hi,
You have done a great job with the web site.
We are preparing a patient information booklet on Trabeculectomy for our glaucoma clinic. This is just for helping our patients and without any personal financial interest.
I will be most grateful if you would allow us to use one of the diagram on trabeculectomy for the same.
Please will you let us know.
Kindest regards
Dr Akash Raj
Russells Hall Hospital, Dudley, UK
Hi,
You have done a great job with the web site.
We are preparing a patient information booklet on Trabeculectomy for our glaucoma clinic. This is just for helping our patients and without any personal financial interest.
I will be most grateful if you would allow us to use one of the diagram on trabeculectomy for the same.
Please will you let us know.
Kindest regards
Dr Akash Raj
Russells Hall Hospital, Dudley, UK
———————————————————–
thank you for this informative site
ΩK.. βΨ RΣΔDIΠG THIS IT’S SΩUΠDS LIKΣ I HΔVΣ ΩPΣΠ ΔΠGLΣ GLΔUCΩMΔ.CΔLLIΠG QUΣSTIΩΠ I HΔVΣ THΔT ωΔSΠ’T DISCUSS ΣD ΩΠ MΨ SΣΔRCH.ωHΨ IS IS ωHΣΠ I TΔKΣ PICTURΣS MΨ ΣΨΣ THΔT IS IΠҒΣCTΣD IT CΩMΣS ΩUT ωITH PUPIL RΣD. “JUST 1 ΣΨΣ ” IT’S ΣMβΔRRΔSSIΠG ΔΠD UΠCΩMҒΩRTΔβLΣ TΩ DΣΔL
Pls sir my mum have glacoma pls d hospital we went to for sugery d doc said they cannot carry out sugery on her eye we are very confuse now sir cos am not happy wit my mums condition i want to know wether there is a glass that my mum can use to make her see clearly whenever she put it on pls i am luking forward to hear positively from u sir thanks
Optic disc may be sinking in glaucoma, not cupping. Sinking of the disc implies a mechanical failure. As a result of sinking disc the pre-laminar nerve diners are stretched and then severed. Thinning of the RNFL, selective destruction of arcuate fibers in the early stage of glaucoma and histology of end-stage glaucomatous disc of empty bean-pot suggest that the nerve fibers are being severed, not atrophied in glaucoma. Glaucoma may not be optic disc neuropathy but an optic disc axotomy.
I HAVE BEEN DI8AG. W/ IRIS TRANSILLUMINATION W/ ENDOTHELIAL PIGMENTATION. DR. TOOK A VIDEO OF, I WILL GO BACK TO HIM IN A MONTH OR SO TO SEE HOW THIS IS PROGRESSING. FOR YEARS I HAVE BATTLED FIBRO -MY FATHER (BECHETS DISEASE)(LIVING 76)& HIS TWIN SISTER(PASSED AT 43) (HISTOPLASMOSIS/BECHETS) WHAT IF ANYTHING CAN I DO TO KEEP MY SIGHT, THE PROGRESSION HAS BEEN RAPID OVER LAST 4 YRS, ALTHOUGH just Diag., did have Lasik when I was 19.
I BECOME A BIG FAN OF YOUR
YOU HAVE PRESENTED MOST WONDERFUL & NATURAL WAY OF OPTHALMOLOGY UNDERSTANDIG
Very welll done brother
Saw ur video on anatomy of eye n 4 d 1st time i understood it completely
Nicely done
Amazing resource thank you for your hard work!
Thank you so much for doing this! I’ve started my residency one month ago and this book has been my best friend so far! I feel more comfortable and integrated because of it!
Corneal thickness is 540 micrometers, not nanometers
You are correct. I’ve updated the text (at least on the site … I’ll have to fix the print/pdf with future release). Thanks!
This is an amazing resource! And the fact that you have made it free speaks volumes about your character and passion for medical education! THANK YOU!