Chapter 10: Introduction to the lens and cataract surgery
If you’re rotating through an ophthalmology department, you’re going to be dragged into surgery at some point. Cataract surgery is our signature operation, so it’s worthwhile to familiarize yourself with basic lens anatomy and surgical goals.
The eye is the most amazing organ in the human body, and the lens is one of the most impressive structures within it! Not only is the lens the densest tissue in the body (highest protein content and lowest water percentage) it also remains optically clear for years despite constant bombardment by light radiation. The lens can even use it’s mighty-morphing transformer powers to change shape and thus it’s focusing power! Not bad, eh?
Some Cataract Terminology
Phakic: When you have your natural lens
Psudophakic eye: When a cataract is replaced with an artificial lens
Aphakik eye: When a cataract is removed but isn’t replaced.
Lens Anatomy
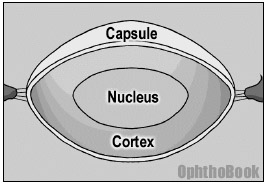 We can’t go any further in our discussion without first describing the anatomy of the lens and how it sits in the eye. When conceptualizing the structure of the lens, you may find it useful to think of it like a yummy peanut M&M candy. Thus, there is an outer capsule like a “hard candy shell” that surrounds the lens. Inside you’ll find the chocolate layer (the lens cortex) and the inner nut (the hard lens nucleus). These three layers are clear, of course, but that’s the general layout.
We can’t go any further in our discussion without first describing the anatomy of the lens and how it sits in the eye. When conceptualizing the structure of the lens, you may find it useful to think of it like a yummy peanut M&M candy. Thus, there is an outer capsule like a “hard candy shell” that surrounds the lens. Inside you’ll find the chocolate layer (the lens cortex) and the inner nut (the hard lens nucleus). These three layers are clear, of course, but that’s the general layout.
Cataracts can form at different layers within the lens, and the location can give you clues to the causative insult and explain specific visual complaints. The lens layers become even more relevant during surgery – with cataract extraction, we tear a round hole through the anterior capsule, suck out the cortex and nucleus (the chocolate and the peanut), and inject a prosthetic lens into the remaining capsular bag.
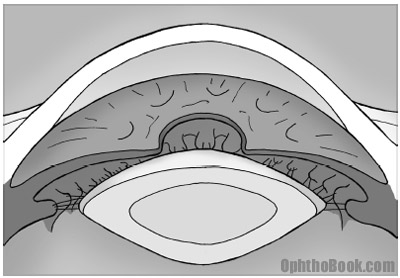
Now, we know the structure of the lens and we know the lens sits behind the iris … but what keeps the lens from falling into the back of the eye? The lens is actually suspended behind the iris by zonular fibers. These zonules attach at the equator of the lens like trampoline springs and attach the lens to the surrounding ciliary body. The ciliary body is a ring of muscle sitting behind the iris. Trauma and surgical mishaps can break the zonules and cause the lens to de-center or even fall into the back of the eye.
Accommodation
Now, I just said that the lens is suspended by spoke-like zonules to the ciliary body. But what is this mysterious ciliary body? The ciliary body is a ring of muscle that sits directly underneath the iris. You can’t see it directly by standard exam without using mirrors, but this ciliary body is important for two reasons: it produces the aqueous fluid that nourishes the eye and it controls lens focusing.
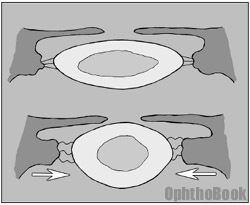 The ciliary muscle can be thought of as a camera diaphragm, or if you prefer a more entertaining description, a sphincter muscle. When this sphincter contracts, the central “hole” gets smaller causing the zonular “springs” to relax. With zonular relaxation, the lens relaxes and gets rounder. This rounding makes the lens more powerful and allows you to read close-up.
The ciliary muscle can be thought of as a camera diaphragm, or if you prefer a more entertaining description, a sphincter muscle. When this sphincter contracts, the central “hole” gets smaller causing the zonular “springs” to relax. With zonular relaxation, the lens relaxes and gets rounder. This rounding makes the lens more powerful and allows you to read close-up.
Unfortunately, as we age our lens becomes harder and does not “relax” into a sphere very well, no matter how hard the ciliary body contracts. This loss of lens accommodation is called presbyopia and explains why we need the extra power of bifocals to read after the age of 40.
Fun Fact!
Ever wondered how those “blue blocker” sunglasses are supposed to improve vision? You know, those yellow tinted-glasses that sport enthusiasts and hunters wear?
They work because all lens systems, including the eye, suffer from some degree of chromatic aberration. This occurs because some wavelengths of light are bent more when going through a lens or prism system. Red light is bent the least, so the color red tends to focus slightly behind the retina, while blue light bends more, thus focuses in front of the retina. This creates a mild blur because not all colors can be perfectly focused at the same time.
Yellow-tinted glasses only allow certain wavelengths to pass through. This eliminates chromatic aberration and the image appears sharper.
Cataract Types and Mechanism:
The lens begins as a clear magnifying glass inside your eye, but with time can opacify. Most cataracts are of idiopathic etiology, though there are many associated conditions that lead to both congenital and environmentally induced lens opacities. Here is a short summary of the important cataract types:
Nuclear sclerotic cataracts
NSCs are the most common type of cataract and many consider them to be a normal maturation of the lens. Over time, the lens becomes larger and brunescent (yellow or brown) especially in the denser central nucleus. If this process goes on long enough the opacity eventually leads to visual obstruction and problems with glare. The lens can become so big that it pushes the iris forward, placing the patient at increased risk for angle closure glaucoma.
With far-advanced cataracts the middle cortical layer (the chocolate layer) can liquefy and become milky white and the nucleus layer (the central peanut) gets hard and falls to the bottom of the capsular bag. These end-stage “Morgagnian cataracts” are rarely seen in this country and are particularly hard to remove at surgery.
Some patients with nuclear sclerotic cataracts will develop so called “second sight” where it seems like the vision improves. This is because the round cataract lens is more powerful and offsets the coexisting presbyopia allowing older patients to read better. Their vision really hasn’t really improved, it’s just that their cataracts are working like weak bifocals inside their eyes.
Posterior Subcapsular Cataract:
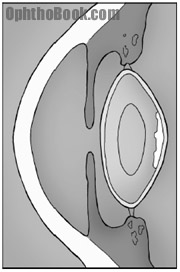 The PSC cataract forms on the back of the lens, on the inner surface of the posterior capsule bag. These cataracts tend to occur in patients on steroids, with diabetes, and those with history of ocular inflammation. The opacity looks like breadcrumbs or sand sprinkled onto the back of the lens. This posterior location creates significant vision difficulty despite appearing innocuous on slit-lamp exam. PSC cataracts are quite common, and often occur in conjunction with some degree of NSC.
The PSC cataract forms on the back of the lens, on the inner surface of the posterior capsule bag. These cataracts tend to occur in patients on steroids, with diabetes, and those with history of ocular inflammation. The opacity looks like breadcrumbs or sand sprinkled onto the back of the lens. This posterior location creates significant vision difficulty despite appearing innocuous on slit-lamp exam. PSC cataracts are quite common, and often occur in conjunction with some degree of NSC.
Posterior versus Anterior located cataracts.
Posterior cataracts cause more visual complaints than anterior cataracts. This is because of the optics of the eye. Advanced optics are beyond the scope of this book. Keep in mind, though, that the eye has an overall refractive power of approximately 60 diopters (40 from the cornea, and 20 from the lens). If you simplify the eye to a single 60-diopter lens system, the important “nodal point” of this system is near the back of the lens.
The closer you get to this nodal point, a greater number of light rays will be affected. Thus, small PSC cataracts are more significant than larger anterior cataracts.
Congenital Cataracts:
Lens opacities in children are of concern because they can mask deadly disease (remember the differential for leukocoria from the pediatric chapter?) but also because they are highly amblyogenic.
Cataracts in the newborn can be idiopathic or inherited. If small or anteriorly located, they may be visually insignificant. However, when approaching a leukocoric pupil, you should first rule out potentially deadly disease. This includes cataract masqueraders like retinoblastoma, and deadly causes of cataract like the TORCH infections and galactosemia.
A true cataract needs to be removed quickly, usually within the first two months of life, because they are highly amblyogenic. Cataract surgery is challenging in this age-group as children have impressive inflammatory responses and are not easy to examine pre- and post-operatively. After taking the cataract out, you don’t implant a prosthetic implant in newborns, but wait a few years because their eyes are still growing. The family must deal with powerful aphakic glasses or contact lens placement until the child is old enough for the secondary lens implantation.
Traumatic Cataract:
A cataract can form after blunt or penetrating injuries to the eye. When the outer lens capsule breaks, the inner lens swells with water and turns white. These injuries typically occur in young men and the lenses are very soft and easy to suck out. Removal and implant placement can be complicated, though, as the blunt force often tears the zonular support. If the lens is barely hanging in position, it may be safer to consult a retina specialist to remove the lens from behind (a pars plana approach) to keep the lens from falling back into the eye.
Lens Nutrition and Glucose metabolism
The cells that make up the adult lens have no innervation or blood supply, and thus derive their nutrition entirely from the surrounding aqueous fluid. Because of this low O2 tension, these lens cells survive almost entirely on glycolysis.
Poorly controlled diabetics can have very high levels of glucose. If high enough, the lens metabolism can shunt down a sorbital pathway. Sorbital buildup in the lens then creates an osmotic swelling of the lens with resulting refractive changes!
If a diabetic patient complains of episodic blurring vision, find out what their glucose has been running. If it has been high recently, don’t prescribe glasses, as their prescription may still be changing from lens swelling.
Posterior Capsular Opacification (PCO):
A posterior capsule opacification isn’t a true cataract, but an “after cataract” that forms after a cataract surgery. I’ll be talking about the cataract surgery technique in a second, but basically, we suck out the cortex and nucleus (the chocolate and the peanut) and inject a new lens into the remaining capsule (the hard candy shell).
Residual lens epithelial cells are left behind after surgery. These orphaned epithelial cells get confused (and lonely) and can migrate along the back surface of the implant and opacify the posterior capsular bag.
This is a common occurrence and fortunately is easily treated in clinic with a laser. The YAG laser is used to blast a hole in the posterior capsule. We don’t break a large hole, as you don’t want the implant to fall into the back of the eye, but one big enough to clear the visual access. This is known as a YAG capsulotomy.
One more topic … Lens Dislocations
As already mentioned, a lens can dislocate from traumatic force (such as a punch to the eye). It can also dislocate because of inherited diseases that affect zonular strength. The two major causes of hereditary lens dislocation are Marfan’s syndrome and homocystinurea.
Marfan’s disease is an autosomal dominant disease of fibrillin. These patients have tall body habitus, arachnodactyle and can have lens subluxation with the lens dislocating upwards. This can create large astigmatism as the patient is looking through the edge of the lens, and may eventually require cataract extraction.
FUN FACT:
Some historians believe that Abraham Lincoln may have
had Marfans syndrome.
Homocystinurea is an autosomally recessive heriditary disorder that results in an absence of cystathionine B-synthetase. This enzyme causes the conversion of homocysteine to cystathionine. These patients have a marfanoid habitus, arachnodactyly, and there is a 50% incidence of mental retardation. The lens zonules are largely composed of cysteine, and without good cysteine, the zonules become brittle and can break. The majority of these patients develop downward lens dislocations. They also have poor peripheral circulation and are subject to thromboembolic events under general anesthesia.
Is the patient ripe for surgery?
Most people over 50 have some degree of cataract in their lens. The question then becomes “should you have surgery or not.” This is not always a clear-cut choice: you can be amazed at the dense cataracts that patients are still able to see through, and conversely, the seemingly “wimpy cataract” that causes major visual complaints.
There is a saying in ophthalmology, “If you can see in, than the patient can see out.” That is to say, if you can see the retina clearly with your ophthalmoscope, it is likely that the patient can see clearly through their lens. More objectively, we generally use 20/50 as a cut-off for surgery as this is the minimal driving acuity in most states, but patients have different visual needs. A visual acuity of 20/30 is not acceptable for a young commercial airline pilot. Conversely, potentially life-threatening anesthesia might not be necessary for a 20/70 nursing home patient who likes jazz music and is happy with his vision.
Acuity isn’t everything:
 One big complaint that people have is glare. In the dark a patient may see fine. But have them drive into the sun or at night with car headlights coming at them, and they become blinded by the scattering of light through their hazy lens. Many patients tell us that they no longer drive at night. We can test glare in the clinic by checking vision while shining a light in the eye. Also, you can more formally test glare with the BAT (brightness acuity tester) device. This is a light-bulb illuminated hemisphere with a view hole that induces glare.
One big complaint that people have is glare. In the dark a patient may see fine. But have them drive into the sun or at night with car headlights coming at them, and they become blinded by the scattering of light through their hazy lens. Many patients tell us that they no longer drive at night. We can test glare in the clinic by checking vision while shining a light in the eye. Also, you can more formally test glare with the BAT (brightness acuity tester) device. This is a light-bulb illuminated hemisphere with a view hole that induces glare.
Another indication for surgery is the presence of underlying retinal disease such as advanced diabetic retinopathy. If a cataract interferes with careful fundus examination or laser treatment, the lens needs to come out.
Who decides?
Ultimately, it’s your patient’s decision whether to have surgery. In an ideal world without operative complications everyone should have cataract surgery as soon as the vision drops to 20/25. Unfortunately, bad things can happen in surgery, and patients have to decide if they’re vision is affecting their life enough to take the risk of surgery. Our job is to educate and inform our patients about these risks and about their surgical options.
Cataract Surgery – A historical prospective:
In Egyptian times, cataract surgery was a primitive affair. Eye “surgeons” would take a sharp needle and shove it into the eye to rip the lens from it’s zonular support and allow it to fall into the back of the eye. This technique, called “couching,” clears the visual axis, because the lens is now bouncing around in the bottom of the eye. Patients had terrible vision after this (with approximately 20 diopters of hyperopia) but back in those days of ultra-dense cataracts, this was an improvement allowing these early patients to see basic shapes, such as the outline of the pyramids and perhaps their camel.
During World War 2, fighter pilots suffered from penetrating eye injuries when fragments of their Plexiglas cockpits exploded. Eye doctors of that era found that the eye seems to tolerate plastic, thus spawning the idea of using plastics to create intraocular lens implants to replace the natural lens.
Cataract implants have evolved since then. Now we have lenses made of PMMA plastic, acrylic, and silicone. These implants can be folded through smaller incisions and placed in different positions inside the eye – in the capsule, behind the iris in the “sulcus,” or even sitting on top of the iris in the anterior chamber.
Preoperative measurements: how to choose your implant power?
Our goal in cataract surgery is to put the ideal power intraocular lens into the eye such that the patient won’t need additional glasses for viewing distant objects. This is not always an easy task, as everyone’s eyes are different and minor anterior-posterior shifts in the lens placement will severely affect the end refraction. There are many formulas designed from both lens theory and regression analysis to help you choose the correct power lens. We won’t be going over these formulas, but keep in mind that we need to measure two things to come up with the right prescription for the implant:
a. The corneal curvature: Remember that the cornea-air interface actually performs the majority of the refractive power of the eye. The cornea performs approximately 40-diopters of refraction, while the lens makes up the last 20-diopters. A person with a powerful cornea will need a less powerful lens. We measure the curvature of the cornea with a keratometer.
b. The length of the eye: The shorter the eye, the more powerful lens you’ll need to focus images onto the retina. We measure this with the A-scan mode of a hand-held ultrasound.
Cataract Surgery – How to Do it!
Cataract surgery is easy in concept, but actually performing this surgery is challenging as you’re working under a surgical microscope with delicate ocular structures.
The Steps:
There are many steps to cataract extraction, and many ways to go about it – everyone has their own combination of machine settings, viscoelastics, irrigating fluids, and preferred instruments. Essentially, you can break down the cataract surgery into a few steps:
1. Anesthesia
Dilate the pupil, prep, and anesthetize the eye. Anesthetic can be given with simple topical eyedrops like tetracaine. We can also perform a retrobulbar block by injecting lidocaine/bupivicane into the retrobulbar muscle cone to knock out sensation through V1, and eye movement by knocking out CN3 and CN6. The trochlear nerve (CN4) actually runs outside the muscle cone, so you can see some residual eye torsion movement after the block. If you’ve never seen a retrobulbar block, you’re in for a treat (it can look gruesome the first time).
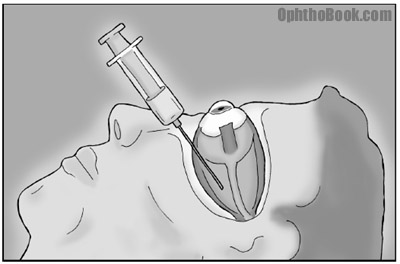
2. Enter the eye
The main surgical entry site can be performed several ways. You can enter the eye by cutting through the cornea, or you can spend more time tunneling in from the sclera. A clear-cornea incision is fastest, while the scleral tunnel takes longer but is easier to extend if you run into surgical complications.

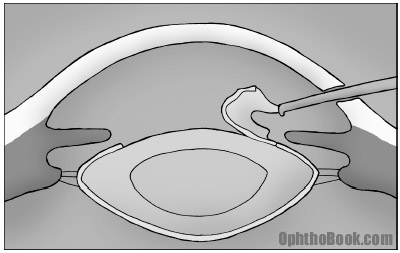
3. Capsulorhexis
To get the lens out you need to tear a hole in the anterior capsule (hard candy shell) of the lens. This step is important to get right, because if the rhexis is too small, it will make cortex and nucleus removal harder. Also, the outer capsule you are tearing is finicky and can tear incorrectly, with a rip extending radially outwards to the equator (not good). If you lose your capsule, you can lose pieces of lens into the back of the eye. Poor capsular support also makes implant placement that much harder.
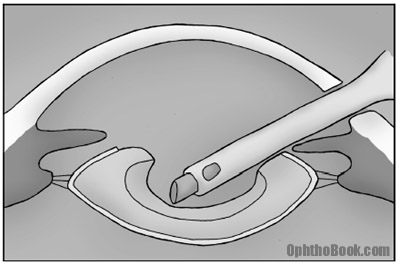
4. Phacoemulsify
We use an instrument called the phaco handpiece to carve up the lens nucleus. This machine oscillates at ultrasonic speeds and allows us to groove ridges into the lens. After grooving, the lens can be broken into pie-pieces and eaten up one-by-one.
5. Cortical removal
After removing the inner nucleus, we can remove the residual cortex (the middle chocolate layer) of the lens. This cortex is soft but wants to stick to the capsular bag. You don’t want to leave too much, as it will cause inflammation and can cause “after cataracts” (posterior capsule opacification). We strip this with suction and vacuum it out.
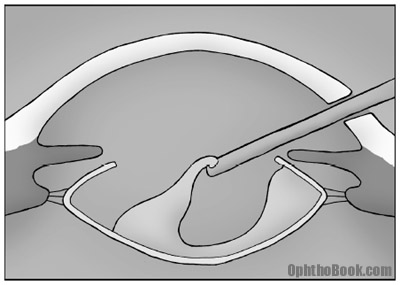
You need to be careful with your posterior capsule during this cleanup. The surgeon tries to maintain the posterior capsule for a couple of reasons – not only does it create a support structure for the new lens, but it maintains the barrier between the anterior and posterior chambers, keeping the jelly-like vitreous from squeezing into the anterior chamber.
6. Insert the lens
We usually use a foldable lens that can be injected directly into the bag. If we’ve lost capsular support (for example, we managed to break the posterior capsule during phaco or cortex removal), the lens can be placed on top of the entire capsular bag, right behind the iris. If support for this sulcus placement is questionable (i.e. you’ve had a LOT of complications with the case), a lens can be placed in the anterior chamber on top of the iris, or sutured to the back surface of the iris (tricky).
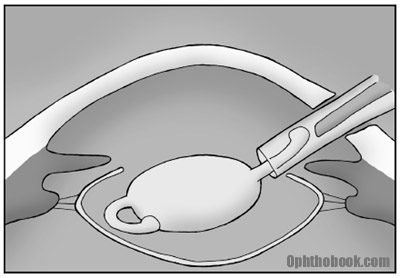
7. Close up
You now close the eye. Many small incision corneal wounds are self-sealing, but some require closure with 10-0 nylon suture that will eventually biodegrade.
8. Postop care:
Immediately after surgery, antibiotics are dropped and a shield is placed over the eye. The patient is then seen the next day and will use antibiotic drops and a steroid drop to decrease inflammation.
Conclusion: Cataract surgery is not easy
Almost every ophthalmologist performs cataract surgery, so there is a tendency to view this as a simple procedure that only takes a few minutes. Some cataract cowboys are able to perform an extraction in ten minutes and may even downplay the risk.
The reality is that cataract surgery is very difficult. The lens is mostly clear, floating in clear aqueous, supported by a microns-thin clear capsule that wants to tear. The patient is usually awake, so any small movement such as a cough or simple head adjustment looks like an earthquake under the microscope. Cataract extraction involves many steps, and early mishaps at the beginning of the case cascade and make the later steps that much more difficult.
Look at it this way: any surgery that takes over 100 operations to develop basic proficiency has got to be tough. Cataract surgery is like flying an airplane … it takes many years of training, screening, certification, accreditation to be accomplished pilot, and most flights are uneventful. But you want a qualified person behind the wheel when you hit turbulence.
Fortunately, most of the time things go just fine.
PIMP QUESTIONS
1: What does it mean to have a phakic eye or an aphakic eye?
Phakic means that the patient has their original lens. Pseudophakic means that they have a intraocular lens implant. Aphakic means that their lens was removed, but no replacement lens was placed.
2. What are the layers of the lens and what is removed in cataract surgery?
There are three layers to the lens. The outer capsule, the inner nucleus, and a middle cortex … in a configuration like a peanut M&M candy.
3. When you accommodate (look at near objects) do the zonules relax or tighten?
The zonules relax. With accommodation, the spincter-like ciliary body contracts, the zonules relax, and the lens relaxes and becomes rounder (thus more powerful). You’re going to have to think that one out a few times and look at the drawing in this chapter.
4. What are the two functions of the ciliary body?
The ciliary body changes lens shape, allowing fine focusing and accommodation. It also produces aqueous fluid that inflates the anterior chamber and nourishes the avascular lens and cornea.
5. By what mechanism can a diabetic patient have a temporary refractive error?
Too much glucose will switch the lens metabolism from anaerobic glycolosis to a sorbitol pathway. Sorbitol buildup in the lens creates an osmotic swelling that changes the lens power (the round, swollen lens makes images focus in front of the retina, thus the patient is temporarily near-sighted).
6. Why do yellow sunglasses make images seem sharper?
All lens systems have chromatic abberation because the different colors of light bend differently. This means that images don’t focus perfectly on the retina – the blue component focuses slightly in front of the retina, while the red component slightly behind. Tinted glasses limit the spectrum of color that hits the retina, and makes images appear sharper.
7. How soon should a child with a cataract go to surgery?
Soon, as cataracts create a visual deprivation that quickly leads to amblyopia. Some practitioners recommend surgery prior to two months.
8. How can a cataract cause glaucoma?
Many cataracts are large, and this can push the iris forward and predispose to angle closure glaucoma. Also, end-stage cataracts can leak proteins into the aqueous and the resulting inflammatory cells (macrophages) can clog the trabecular meshwork.
9. What measurements must you have to calculate a lens implant power?
You need to know the cornea curvature (because the cornea performs the majority of the eye’s refractive power) and the length of the eye.
10. How much of the lens is removed in typical cataract surgery?
With eye surgery, we create a hole in the anterior capsule and suck out the inner nucleus and cortex.
11. What’s the difference between and PCO and a PSC cataract?
PCO: posterior capsular opacification. This is an “after cataract” that forms on the back surface of the posterior capsule after successful cataract surgery. This opacity can be cleared with a YAG laser.
PSC: posterior subcapsular cataract. This is a cataract that forms on the back subcapsular portion of the lens. These tend to occur more often in diabetics and those on steroids, and tend to be visually significant because of their posterior positition.
12. What does it mean to place a lens “in the sulcus?”
The sulcus is the space between the lens capsule and the back of the iris. If the posterior capsule is torn and can’t support the lens, you can often place a lens on TOP of the entire capsule in this potential space.
13. What drops are given after a cataract surgery?
Usually an antibiotic, such as ciprofloxacin or vigamox. Also, a steroid is given to decrease inflammation.
I have been told that my implanted lens is on an angle and there is a samll possiblity that during replacement of the lens it may slip back in to the viterous humor.
What are the chances of this happening?
What specality should an opthalmologist have to do this surgery?
What is the possibility of permanent damage being done to my eye?
What would have caused this slippage of the lens?
My opthalmologist never mentioned it, but sent me to an optomertrist to have ??? drops for a dilated pupil. The opthalmologist never explained why I needed this and the optometrist would not touch my eye. Please help
Well,
Hard to say given your history. Sounds as if anything could have happened:
1. The implanted lens may be at an “angle” because it tilted as your capsule contracted.
2. One of the haptics of the lens implant may be sitting in the sulcus
3. You may have had some zonular dehiscence with instability of the supporting capsule.
4. You may have had a posterior capsular tear during the first surgery (common)
You should really speak with an ophthalmologist whom you trust to look at your eye under the microscope and give you an opinion and give an opinion on your chances … I’m just guessing.
I will say that this kind of lens tilt can happen after surgery, even after a “perfect surgery” without other complications or difficulties.
As for the chances of a “dropped implant” … this is really a possibility, as a secondary operation is much more complicated and difficult than the primary surgery. If the lens DOES drop, it’s not the end of the world … but you’ll need another surgery by a retina surgeon to remove it.
Like I said, though, you really need to see an eye doctor to examine your eye closely and discuss this with you as it’s almost impossible to guess without seeing the eye.
Hello,
i am a medical student at the end of my second year of medicine.
I recently heard about a new invention in cataract surgery:
Its called the Nanoknife.
Its supposed to drastically reduce capsular opacification after surgery.
Do you know about this?
Thanks
P:S– this page helped me get through my end of rotation exam really well.
hi,
i would like to know the blood sugar cut off level & b.p prior to cataract surgery or any ophthalmic surgery.
thanx
Following a disasterous cataract surgery. (Eyeball filled with debris that required vitrectomy that led to CME) I eventually developed an elliptical iris that allows light to leak past the lens implant and being unfocussed produces haloes and glare. Is this a common complication? The article is good and I liked the style. C. Allan Young
dear all,
i am pharmacist and so interested in cataract surgery. But I have one question: what is the role of viscoelastic substances like(H.A) in cataract surgery??
thanks
Editors Note: The viscoelastics used in surgery serves two purposes. The first, is to provide protection to delicate structures inside the eye – primarily, the endothelial surface of the cornea (those endothelial pump cells are very sensitive and don’t regenerate when lost). The second purpose is to keep the anterior chamber deep (keep the eye from ‘deflating’ when removing instruments and inserting the implant).
I would like to know how you determine (about) the preoperative myopia of a patient that had cataract surgery without an implantlens, and who needs a refraction of +3 in his glasses.
Could you also include a formula? Thanks!
( I like your website a lot!!)
And can you help me with this: it is generally assumed that high(er) myopia correlates with low(er) vision. Is this true? Can’t find anything good about it. Thanks!
nice work
My mom recenty had eye surgery to have cataracs removed.
In a recent surgery, her doctor told her one of her lens in her right was tilted. The doctor said my my mom can see netter than before…but said she may trouble later with sunlight. Should she have another surgery to fix it? Are we doing more harm than good.
I had cataract surgery in June ’08 (IOL implant) and later, in July 09, had retinal reattachment surgery (intraocular surgery with gas bubble)on the same eye for a partially detached retina. The retina had two flap tears that were repaired (cryo and laser). Five months post retinal surgery I have distorted vision in the operated eye: the image I see slants downward, from right to left (sloping horizontal axis of image) and the image tends to be elongated vertically. My IOL was sutured in place at the start of the retinal surgery, I assume as a precautionary measure. Is it possible that my IOL was somehow displaced during my retinal surgery and is causing this vision distortion, or is it more likely that other reasons for the distortion are more likely? Is there a way an ophthomalic exam can determine whether the IOL is in the right place?
Thank you for your assistance. Your web site has been very useful to me.
Steve
Can you please tell me where did you derive the A-constant in the SRK meaurement..
hello,
pls tell me what is the cutoff glucose level before cataract surgery.
my dad(diabetic) had 140 fasting on the morning of surgery.the diabetologist told the nurse on phone to give 4 units of insulin.so he was given…and in an hour,his level came down to 133.his surgery was performed.On the night after surgery..his ocular pressure had increased,he had pain.but mannitol to reduce pressure was gvn only next day…when v reported agin to hospital.and later tht day v came to knw tht diabetologist was denying tht he said the nurse to gv insulin to my dad.i mean he was like i first said the nurse to give bt then later denied.i dont know y he behaved tht day,isnt 140 more for fasting,n gvn insulin must have only benefitted him na??
Now 3 wks later,when he is facing probs with vision, n also floaters,doc(opthlmologist) has told he has gt retianl swelling,he was gvn tricot inj. n he is on acular eye drops now for 2wks.pls gv info.
thank you vvvvvvvvvvvvvvvvvvvvvvvvvvvvvery much
Thanks for such a wonderful e learning chapter!
Dear Sir,
I’m a 3rd year medical student in India and I’ve an interesting doubt. Can the patient see the operative procedure when done since his Optic nerve is not targeted in Anesthesia?
Thanks again.
Dear Raviteja,
good question – some patients are worried before surgery that they will see large knives and needles moving just in front of their eye. Luckily, the microscope lamp is so strong that the operated patient cannot see any details with the operated eye, and the other eye is covered by sterile cloth. So no need to worry about seeing fearful surgical tools.
On the other hand, if you use subtenonal or (less common today) retrobulbar anesthesia, the optic nerve is blocked and the patient will experience that what vision they have goes away. This might scare them if they are not told in advance that this is normal and that vision will return after the anesthesia.
Best wishes
Bjorn Johansson, associate professor Linkoping University Hospital Sweden
good luke for all student of medical
and best wishes
Thank you!!!! you give important things.
what happens when a lens turns after cataract surgery?
My Ophthalmologist told me today that my lens implant had slipped changing the shape of my eye which resulted in a astigmatism and vision change. Can this be corrected? Did he not put the lens in correctly?
sir i just want to know can we dilate the fundus by using tropicamide while we are using steriod eye drop
My sister recently had cataract surgery. She now can read and see w/o glasses. She had worn glasses since she was 1 yr. old due to “short eye”…Could this type of surgery be done years earlier to spare her needing glasses for all those years?
during eye refraction through retinoscopy sometime scissor reflexes are seen,instead of maridian reflexes,what should proper step for the neuterlization is require.
what happens when the pupil sticks to the replacement lens not allowing the pupil to function
hi
i am a medical student of final year mbbs … we have ophthalmology subject in final year.. you book ,videos and fun part have helped me a lot, and have created a special interest in this subject.. thanks a lot, your work is really appreciable
hi iam a med student from germany and got ophthalmo for my last oral examination… this site helped me out a lot!! makes learning about the eye fun 😀 thanx!!!
Hi…
I am an Optometrist
I dont seems to find any information regarding “Cortical cataracts.”..but runs into them off and on from refering Doctors.
Please help!!
This is my first time to go to your website. I found it informative and educational. I have a question though. I need to have cataract surgery for both eyes and I have two option for the type of IOL: i)lens in one eye that will provide near vision and an IOL in the other eye that will provide distance vision, ii)lens in both eyes that provides distant vision. Can you tell me the pros and cons of the two options? Thank you.
Tim Root: Abul, here is my standard response to patients concerning cataract-monovision:
Monovision is when one eye is set for distance, while the other is set for near. Many contact lens wearers use a “monovision contact prescription” to avoid reading glasses. Most will set their dominant eye for distance and their non-dominant eye set for reading.
While some people (like my mother) tolerate this imbalance well, other people hate monovision. The disparity between their eyes makes them sick to their stomach and unsteady on their feet. The blur at distance ruins their depth perception for driving, and reading becomes a chore because one eye is doing all the work.
Imagine if we set your eyes permanently this way during your surgery. If you don’t like your monovision, it’s going to be hard to change those implants! At least with glasses or contacts we can take them off or pop them out.
If you’ve been using monofocal contacts for years without issue (or your eyes are “naturally” focused this way), then we may purposefully shoot for monovision … but not without discussing this at length during your clinic visit.
In the end, most people prefer to have both eyes in synch with each other. You can always put a contact in one eye for monovision afterwards if you really want it.
My daughter was born with congenital catarracts and had then removed at six months old. The lens capsules were removed during the operation. Can you tell me if there is an artificial lenses that can be implanted that doesn’t require the lens capsule to be present?
Sir,
I had cataract removal and a partial corneal transplant in my right eye in June 2007. I developed high eye pressure the evening after the surgery which was releved the next morning. Otherwise, everything was fine. In the spring of 2009, I had to have a YAG procedure due to clouding of my vision. This was successful. I have again developed clouding in the eye, essentially in the same area as before. Is it possible that I need another YAG? What could cause this to happen? I do have Type II diabetes, but it is controlled.
Thank you,
Elizabeth Maness
I am doing Cataract+IOL surgery audit( biometry) to find out our predict errors and then cutomise our A constsnt, could you please help me where can I find causes for refracive deviation to design my proforms please.
Thank you
Mabruka Azzaruk
After 2 years of lens replacement for cataracts, I woke up this morning, and can see an outline of my new lens ( I believe this is what I am seeing) No pain or pressure. Just disruptive. Any ideas?
Tim Root: Sid, you need call your local ophthalmologist. If you are seeing something round in your vision … it’s probably NOT the implant, but more likely a vitreous detachment. Many people see a round, clear floater (called a Weiss Ring) when the vitreous jelly inside the eye contracts. While vitreous detachments are usually harmless, this is sometimes a harbinger of more serious problems like retinal detachment, so call your ophthalmologist first thing and get that eye checked out today.
dear sir
iam a pilot and have flown since 1993. throughout i had a slightly blured vision in my left eye recently during optalmic eye check up with dilation it was diagnosed that i may be having the coronary cataract or some call as blue dot cataract ,should i continue to fly
i am a 47 yo male,in 1986 i had a tramatic cateract,had an intraicular implant done at the jewel srien clinic at UCLA.all done under workman’s comp. i got a small settlement,alot of grief.10 yrs of dry eye,chemical sensitivity,the worst was the iliments growing like grains of sand.my lawyer was smart and got a judgement for lifetime medical. well its 24 yrs later and the lense has slipped out of its mounting. so it seems to me putting a new lens in asap is the smart thing to to. ..now the foot draging by the ins company.seen 3 specialists that do not want to touch it,seems the groove the lens sits in is eroded away.this is going on a year now and im very frieghtened. i want to go back to jewel stien and have it done despite the risk,which im told is 50/50 of loss of vision.the ins company offered me 15k.i said no.they are asking for a counter offer and termination of the settlement.i now have contracted AIDA and liver failure. so i am over whelmed. hould i settle and whats that worth? what would that link of surgury cost? is fixing the eye doable. please give me your two cents.it woud mean the world to me.sincerely,alan hunter..818 692 1420
hi, My mother inlaw had cataract surgery and during surgery her iris slipped out,had a small tear,fixed they said .This is only happened yesterday.Today she is not seeing that well,I told her to give it a few days since she had the toric lens.I had never heard of the iris slipping out.What is your take on this. Thank you
Dear Sir/Madam,
My name is Les and I am 60 year old male living in Australia. About 12 months ago I began to experience unusually unstable vision. The moment I stayed in the sunlight for about a minute or longer without wearing sunglasses my vision the next day would go blurry and stay that way more or less. Gradually my vision turned to opaque and partially distorted when looking out close or far. A few years ago I got myself a pair of pin hole glasses for reading and it was working just fine. I tried to put them 10 months ago and I have noticed that pin holes this time were turning into white haze. As it is of now I can hardly read wearing them because my vision is dim and kind of distorted and I see through white fog. I went to see an eye doctor and he said that there is some protein layer formed which kind of obstructs the lenses. On top of it if I look at something out there my eyes ache and sting and it is not comfortable at all times. I am forced now to wear sunglasses at all times for the reason mentioned above.
I’d appreciate your comments if you don’t mind.
With regards,
Les
50 year old post-LASIK patient (15 years ago) with great results. Now had aggressive cataracts removed and single distance implants in both eyes. HATE VISION! why is my distance vision soo strange and disorienting even though acuity is 20/50? Have no near or mid vision at all and little depth perception. Peripheral vision on left side poor. Now need progressive eyeglasses full-time and even these do not fix problems. Need YAK in both eyes (1 week post surgery), PRK in left eye and surgeon suggests LASIK again in both eyes. Will this help-is it likely to correct mid and near or at least improve without compromising distance vision? Are piggyback lenses an option? Any other suggestions? My surgeon is not exactly forthcoming with info. Has anyone else experienced such traumatizing vision after catatact removal?
I know a man about 50 years old who lost a lens due to a childhood accident. He’s not been under the care of a doctor since then. Could that lens be replaced after all these years. He was recently diagnosed with diabetes.
hi
my born with cataract, we do the surgery and remove the cataract when he is 2 months old. we use external lens now he is 4 years old. when we do the final operation and implant internal lens. please tell me urgent
Hallo,
My wife Hildegard (84) had her first catarac operation 5 weeks ago. The membrane behind the eye was raptured, resulting in very extensive pain for almost 2 weeks. The surgeon assured her,she will be alright and prescribed eye drops she took every 2 hours during the day, and a guard was over the eye during nights. She seen der doktor twice during the next 4 weeks, and today he revealed that he could not detect the implanted lense and suspected it disappeared in the lower eye. To retrieve it she would have to consult a retina surgeon, or he could ignore the lense and install a replacement without further cost to Hildegard.
The hidden lense would be of no harm to her. She opted for that solution. Hildegard has to see the doctor again in 4 weeks. By then her injured eye should be healed and the procedure could take place. Milder pains are still prevailing, but further eye drops are necessary. She would like to know what the possible outcome oft this misshab
could be. Your professional answer would be very much appreciated.
Hildegard
Hi
I had both lenses removed in the 1970’s due to cataracts and
hve worn thick bifocals and contact lenses since then. Are atificial lenses an option for me?
hi, i am a young man in my early twenties.the problem is my vision is very poor in the dark, do i need to wear some spectacles????
@ignatious Is ur vision poor just in dark or blurred even in day time?? n do you notice glare on seein bright light? n wat abt double vision? n do u c black spots?
With cataract surgery, assume a lens with 31.00 diopter was implanted by one opthamologist and resulting vision was worse than prior to surgery. A second opthamologist was consulted and after testing recommended a 27.00 diopter lens. The eye which has yet to be operated was to be a 34.00 diopter lens per the first opthamologist while the second opthamologist recommended a 30.50 lens. During testing in the office of the first opthamologist they had considerable difficulty in getting measurements and had to adjust the ultrasound apparatus to get the instrument to read. Is it possible this might suggest why the recommendations were so much different? Question – what to do in making a decision about surgery on the second eye?
I had surgery 1 yr. ago & lens was put in wrong. I seem to be looking thru fog. I had Laser treatments on both eyes & now having trouble with my right eye now & no help for left eye. Could I have a second surgery to correct that bad lens or do I have to put up with this forever? It is ruining my
life. I fell recently & crackd a rib because I can’t see properly. Please let me know what you think about a second surgery, so I know what to do. Thanks.
This is my valid email address.
I heard that it is a good idea not to do lots of reading within first week after cataract surgery. The rapid changes in the size of pupils can result in IOL sticking to it. Bit it is ok to watch TV at a distance. Is this correct?
I had cataract surgery and everything was good and seeing good. went back to the doctor and she said she would do a yag lazer on the eye and thats all you would ever have to do. after doing it i now see hugh starburst whenever car lights or other lights hit it. what went wrong? the doctor said there is nothing she can do about it.
What could be the reason if blood comes out during laser operated cataract eye surgery?
I recently had cataract surgery in left eye and now i can see the rim of the new lens as a dark curvy image,this is very uncomfortable and irritating: my question can the lens be taken back out and replaced with another one?
thaaaaaaaaaaaaaaanks
My mum has just had her cataract on her first eye today, but they took out her old lens but could not put a new one in. She therefore has to wait 2 weeks and go back to outpatients to see the consultant. Does anyone know why they could not put a new lens in? and why would she have to wait for 2 weeks?
Hi
Due to a severe blow to the side of my head, the left eye lens is almost detached and wobbly. The surgeon says there is a high risk that the lens will fall back into the eye on attempted removal for replacement with a lens implant as at present the ‘bag’ behind the lens is falling over the upper edge of the lens.
Although I can ‘see’ with the eye, vision is extremely poor and out of sync with the other eye.
I am told that vision with this eye will never be as good as before the accident.
What are the risks in this type of surgery?
Kind regards
Keith Harris
editor
newsmedianews.com
Your article is very informative. Thanks for the good works.
In the cataract surgery procedure, a small top part of the anterior was ripped or tear to get access into the len itself. After the IOL was injected and placed firmly inside the capsule, what happen to the ripped part? Would it grow or heal back again to cover the len?
Thanks.
A K Teo
Singapore
i had cataract surgery 4 weeks ago. there was a complication due to the cornea being soft. i had stitches, but, i was always myopic and now, i can see very clearly near, but far objects are distorted (unable to focus clearly) the surgeon says it is astigmatism, WHICH I NEVER HAD BEFORE! What am I to do before he does the right eye when i cannot see out of the left?
I am a medical student, in my 4th year
this topic helped me so much in my finals
I love ophthalmology more and moreee :))
wonderful guide.
Would like u to put some clips on SICS for the benefit of us in the third world.
Exceptional guide for trainees
On my lens implant card s there are two numbers used in the power identification part. Right has 22.0 D and left has a 21.5 D . What do these numbers mean?
First surgery with Starr implant on distance lens for monovision resulted in 20/20 the day after surgery, but the lens vaulted a month later (an unusual outcome, I’m told), which meant wearing a -1.25 strength contact lens to be able to see clearly. During second surgery three months later, a different surgeon rotated the Starr lens nearly 360 degrees to correct the vaulting problem. It didn’t. Now he wants to perform YAG Laser on capsule so lens will “relax.” However, I fear complications (such as later retinal detachment). The options? YAG laser or wearing a -1.0 soft contact the rest of my life to correct to 20/20. (The latter doesn’t put me off, since I’ve worn contacts for many years with no problems.) Seeking input from other professionals about these two options.
Hi Dr. Root!
I’m a final year med student from Germany, and after already using your brilliant site to study for my ophtho exam a few years back, I’m now returning to revise for my state exams. Your humorous approach to education is truly refreshing between reading so many dry books. Thank you for that, I wish there were more professors like you.
Here are some things I noticed when reading this chapter, for your consideration:
“The lens can even use it’s mighty-morphing transformer powers to change shape and thus it’s focusing power!”
– I’m not a native speaker, but I’m fairly sure it should be ‘its’ both times – not ‘it’s’! I’m a grammar nerd, okay.
You name the nuclear cataract as the most common form – in my German book it’s only second place (with ~30% of cases) after the cortical cataract (~50% of cases in the senile category). Maybe this differs in different populations?!
Many thanks for your hard work,
Sophie
hello…i was diognized with coates disease at the age of 4 in my left eye and had already loss my vision completely in that eye.years after cataract developed on that eye…it has been almost 12 years now the cataract is mature…my doctor never wanted to remove it because he thought my eye would become dry…bt before 4-5 months ago i had convinced him to operate to remove the cataract..bt during the surgery he said that the cataract is too hard and would need to use cutter that can damage my eye….so it was just left there…i would like to know if something could be done…is performing surgery to that eye can damage it…or maybe i should consult some more experienced doctor…and what will happen if i let the cataract unremoved…any kind of suggestion would be helpful..thank you
I had cataract surgery (simple IOL) with corneal loosening for astigmatism yesterday. I had a bleeder in the anterior chamber. The bleeding appears to have stopped and there is no apparent increase in interocular pressure. Today it was 12. How common is this situation and what are the percentages for a positive outcome. I am now taking Besivance, Prednisolone and Ketorolac drops in the affected eye. The surgeon appears competent and performs over 75 cataracts surgeries per month. I do have severe myopia (-11)
Enjoyed this article as well as the different scenarios, it is helping me to learn.
I had a cataract removed earlier this month, I am 54, this was the result of an horse related injury to this eye years ago.
My vision is worse and was told that the zonule fibers are weak and unable to sustain the lense and being fitted for a contact.
In reading over this information as well as being a Medical Massage Therapist, I am wondering what I can do to help my eye strenghten. I look at muscles in a different way and understand their healing. Have added eye exercises (I just found this out yesterday) working acupuncuture points, added larger doses of Magnesuim as well as a repair tendon/ligament formula (Chinese formula) to see if this would help to strengthen the fibrillin.
It is so close to the surgery my Doctor, who is amazing, said there may be a possibility of it strengthening.
Any other suggestions? I feel very fortunate that if worse comes to worse, I can resort to a contact.
I am thrilled and awed at this procedure and the human body.
Thank You,
H. Dennis.
I had catarac surgery and crystal lens implanted. It has been nearly 3 mos now but I continue to see some colors differently. I was a decorator, wardrobe consultant prior and I KNOW colors. However, now some of the same things I KNEW (and everyone else knows) are black, look navy blue. Also, some colors look plum when, in fact, they are actually gray or charcoal. Have you heard of this and will it correct itself or is there any way to fix it?
Sharon,
Colors are a subjective phenomena. Not all people see the same shade of a color or even the same color when they look at an object. Having been a decorator you must have come across what seemed to be terrible color schemes to you, but not the your clients who were not seeing the same colors you.
There are some physical characteristics of the human body that are exactly the same with all humans but color vision is not one of them. As we age the crystalline lens turns yellowish. After your operation you are seeing through the same eyes as a child and the colors are enhanced. A simple test for you take is to look at the blue flame of a gas fire and you will see a purple haze around it which you may never have seen before but has always been there.
Hi there,
I am a 40 year old man. Some 10 or so years ago I had a trauma to my right eye, this led to a detached retina.
The treatment originally was stitching the retina back in place, this didn’t work so the next treatment was ‘posturing’ with a gas bubble in the eyeball. This too was unsuccessful and the final treatment was to remove the viscous fluid and replace it with a heavier fluid which held the retina in place while it healed.
This appeared to be successful but the liquid led to a cataract being formed.
I had a lens replacement. It seems to be a fixed focus, mid range. In a dark room with a light source behind the person I am talking to, the lens can appear to be reflecting light and shimmering.
The vision in my left eye is perfect.
My iris in the right eye doesn’t seem to change size when bright light is introduced. Is a fixed iris the result of the lens replacement? Am I more susceptible to bright lights? Is this a medical fact or is it dependent on aftercare and treatment?
Many thanks in advance
Great Site! Very helpful for my MS3 ophthalmology rotation. Thank you for making it available online for free.
Hi, Had caataract surgery about 6 years ago with no problems. Drove to CA and when I went through two tunnels, I thought the lights were out in them because I could not see even the car in front of me, Thank God that the tunnel wasn’t that long. Is this common with implanted lens or were the lights really off?
If it is my eyes, don’t know how I can drive long distances again for fear of this happening again.
Thank you very much
Charlotte
Tim Root: Charlotte, your experience does not sound like a cataract or lens issue, but rather a slowness of dark adaptation. This could be a normal aging change, or could be the sign of retinal problems. For example, I often hear this complaint from people with macular degeneration. I recommend seeing your eye doctor to look into this.
I had a lens implant put in in January. Then a month later my retina detached. Retina surgeon said the implant was loose and I was wondering if this could have caused my retina to detach.
There is some thought as to adding a post-op topical NSAID (ketorolac) esp with diabetics to reduce the possibility of CME…?
Hi,
My 15 month old son has just been diagnosed with ectopia lentis in both eyes which he has had since birth. He doesn’t seem to have any other symptoms which would indicate any syndrome but he is currently being checked. The doctor has referred him to have regular checks, is it more than likely that he will need surgery to remove the dislocated lense and to put an artificial one in? If so what ramifications does this have? Will he still need glasses or contacts? Will he be able to play sport and in particular contact sport if he wishes when he is older? Or will we have to sit him out of sport????
His lenses are currently too high, what will he be seeing? He doesn’t seem to have much of a problem with his sight but he is too young to tell. When he does have surgery, will that be it, or will he be having monthly checks for the rest of his life?
In the 3rd paragraph, there is a typo: Psudophakic…
…which should be Pseudophakic.
hi, i m 36 years old, i m remothoid artritis patient. i was take joint injection take deltason medicine oral. six months i was not seeing anything clear, doctor said i need facco surjery, my present vision is 6/12, after 8/12 then i will ready to surjery ,now continue giving eye drops . plz tell me what lens i will take both two distance lens or only far distance lens or both. when i will do my surjery, plz tell me what should i do.
My accommodating lens failed–I had 20/20 for 4 weeks and then in dropped to 20/60. Went to another specialist and he talked about piggybacking another lens on. What would be the success of this?
I ad cataract surgery on my right eye five months ago and had a ReStor lens implanted. I have had difficulty since with stinging in the eye and aching around the eye socket. I have been to several ophthomalogists but none can suggest anything but eye drops which do help with the stinging but not the pain on the outer edge of the socket. Any ideas what can be done? Could the wrong size lens have been implanted?
Not allergic to drops by any chance.
Hi your book is very educative .am an ophthalmic nurse. I want to know the post operative management and complications after surgery. Thanks hope to hear from you
I recently had cataract surgery on my right eye. They put in a crystal lens.
It’s been several weeks now and although I can see up close I cannot see in the distance. My doctor told me that the crystal lens moved forward and they will now have to do lasik.
I don’t understand how the crystal lens can move forward if it is been embedded in my eye.
Should I go to another optometrist for another opinion?
Hello, my 1 year old grandson has ectopia lentis both eyes, his pupils are also misaligned. He is to have lens replacement, how often will he require this as he grows? and how will he accomodate if zonules are ‘snapped/weak? He has had blood tests to confirm or rule out Marfans or other cause but not had results yet. He is bright knows what things are and if instructed to pick up say, ball he will crawl to it and pick it up. he is able to see the tiniest things and watches tv and recognises people no problems. My daughter had obstetric cholestasis during her pregnancy and traumatic forceps delivery could this have any bearing? Any information you have would be greatly appreciated, thankyou Julie.
Hello,I am 70.I had cataract removed and they put Lens.But still I need long sight pair of glasses.Could I have second surgery to get lens that does not need long sight glasses.
Thank You.
Amir
Hi. i would like to ask. my brother had an eye operation 12 years back and now he says the eye does not see. i would like to know if is it possible for him to do another eye operation so that the eye can work again. Im so hurt about this im worried about this thing.
Hello, my 65 year old mum had successful cataract surgery 3 years ago on both eyes; she had severe myopia and astigmatism. Recently one of the replacement lenses came loose and has been floating around in her eye. The surgeon told her that, due to her severe eye conditions, he cannot redo the surgery and the only option is to remove the lens altogether without replacing it. Will she be able to see afterwards? I’m worried about her. Thanks!
I am 67 and had cataract surgery three weeks ago on my left eye. Before that I was very very nearsighted, corrected by glasses. I chose the lens to see distant objects clearly. Although my vision at long distances is fairly clear there is no distance where I see text as clearly as with my right corrected by glasses. And although I was told that reading glasses will be needed once my vision is fully stabilized I notice that no over-the counter glasses help at all with my reading currently. So why will they work later since shouldn’t I be close to done with the healing by now?
I have delayed doing my right eye as I am concerned that my current healing is not on track. Other problems include:
Contrast seems too sharp. It’s like I’m looking through a lens that is too strong for my acute nearsightedness.
Blues and whites look purplish particularly if bright.
Too light sensitive.
I can’t focus both eyes together.
Tiny black dots moving across my vision
When looking at text – hazy, blurry, shimmering, sometimes double image (white text on black background.
I saw my opthamalogist last ten days ago and she said my eye is just inflamed and she now has me taking Prednisone drops 8 times a day.
Is something wrong and if so, how can it be fixed.
@WillfromSF Will, I don’t normally answer questions like this on my website (this is more of a medical student teaching project) but I’ll make a couple of comments.
1. Blurry Vision: You’ve had one cataract surgery, but If you haven’t been given glasses, yet … there is no way to know what your vision is going to be. If you can’t see well at “any distance” than you might have some residual astigmatism that only glasses will fix. Here’s a trick … punch a hole in a piece of paper, and try looking through this pinhole around your house. If your vision is markedly improved, then we know that glasses will probably get you that clarity you want.
2. Light Sensitivity: This sounds like some residual inflammation (called iritis) as your eye doctor described. Prednisilone eye drops 8x a day is a LOT … indicating that there is indeed some residual inflammation seen on exam. This is good … as the drops will treat it.
3. Tiny black dots: This is likely vitreous floaters. While usually harmless, they can be a sign of more serious problems like an impending retinal detachment or infection. Mention this to your doctor (who has probably already looked in the back of your eye to check the retina).
Ultimately, your eye condition can ONLY be evaluated by an exam, so don’t take this as medical advice.
Everything your telling me sounds pretty typical, however … while most people have excellent results, some people have more inflammation and take longer to get there. If your symptoms worsen suddenly … see your doctor immediately. If you are unsure of your eye’s progress, then get a second opinion from another doctor in town. Finally, hold off on the second cataract until the first eye is to your liking. Good luck!
I appreciate the very useful info, Dr. Root, as I am really perplexed (as you could tell) about how best to manage the situation in light of getting on with my life. Very helpful.
Question: Can lens replacement surgery be done a second time in same eye?
After having left eye with cataract/lens replacement vision is now only 20/40. This is dominate eye that was to be for distance. Holding off now on second eye though it too needs surgery. Was planning on having that eye for reading. Now trying to figure out what to do next.
Question is can the left eye have another lens replacement to correct vision. Is lens replacement a one time surgery per eye?
If I have right eye done for distance also what is best approach ? How will the eyes even up? Can my right eye have 20/20?
After cataract surgery with a Technis single focus IOL, I see a 1/4 halo with starburst to the top left of my right eye and a 1/4 “glint” to the bottom right at night around lights and glare, also In the day if it is dark inside and bright outside. It makes night driving very distracting and I avoid it. I also randomly see a disturbing moving pattern of silver or grey overlapping trapezoids, triangles? on the peripheral right side of the implant eye. It has been 10 months since my surgery. My doctor says the lens is perfectly placed and that is all. Can you help me understand why I have this problem? It makes me want to have the IOL removed. I was better off with the cataract. Thank you.
I’m a 58-year-old with retinal detachment history both sides, good results, but in my first RLE (next is postponed for the moment) a deficiency of zonules turned up. About 120 degrees of the arc are absent. Surgeon went ahead and implanted anyway, and commented that she also inserted another component (MA68?) to help stabilize. She rejected the capsular ring option for fear of the whole thing falling back into the (vitrectomied) eye. My Qs:
– Will the 120-degrees arc of ‘absent zonules’ increase and and thus destroy the whole thing?
– Is there any surgery that can stabilize the new lens and substitute for deficiency of zonules?
– I ask because having this ‘sword of Damacles’ – it could fall to pieces any moment – is a serious option-blocker and potentially will cause me great harm.
?
Yesterday I had realignment surgery for out of position PC IOL. I am addicted to reading on my iPhone. Could this cause more problems with healing or possible new slipping g out of position of the lens even though I am focusing with the other eye while reading? That is is the recovering eye using the muscles to focus even if eye closed? I’m really scared I will wreck my eye. Drs nurse said it was ok to read on iPhone.
Hello, I had A Yag laser procedure done 3days ago,since then I am experiencing what appears to be a membrane causing a momentry blurry vision, I have a few floaters also,but was told this was normal.Do you think this should be reported to my Opthalmologist?.
I am allergic to PMMA liquid and powder mixture when it gets on my skin. I also can not wear the silicone nose pads as I get extremely red areas under them and I know it is an allergy also. What kind of lens should I have for my cataract surgery? I also have astigmatism. I have been advised to get laser surgery in conjunction with the cataract surgery. Is it a good idea to do both at the same time. Can you do one first? then the other. What complications can I expect.
sir… i am having a black spot on my eye lens…… at the time of seeing it will in front of my eye lens…….. it is disturbing a lot to me-……. so please give any solution
– patient’s remark to Jnnny’s query re her 65-yo ‘mum’ (usage important – presumably a Brit)…
– you have not listed and obviously cannot list all the details on a site like this, but a couple of things are clear:
(i) You need a second opinion. Is your mum going to Moorfields? Private health? Discuss with GP. Unfortunately in the UK, if you are already seeing specialists not in Moorfields, it is difficult to get into Moorfields. But try. And you might get a consultation with the best Private Health surgeon you can find. Good luck – 200 pounds for a half-hour chat. BTW from my own long and at times unhappy – but finally good – experience with NHS eye-surgery staff they have been over the entire range from (a) incompetent to (b) world-best. So you need to find (b).
(ii) you need to become ‘your own expert’, so do lots of googling, esp surgeons’ blogs.
(iii) how good is the fellow eye? Your mum’s overall quality of life over the next 10 years is paramount. At one extreme, 2 years of ‘surgery-and-recup’ misery is bad. On the other, 10 years of bad-vision misery is worse.
(iv) To answer the q of aphakic vision – in principle obviously this is possible with thick spex as was the standard of practice until the 60s when IOLs became universally accepted. But in your mum’s specific case, whether that is an acceptable solution, only you and your mum, with expert help of a specialist, can determine.
(v) Good luck.
Why would I get sunglasses effect in my eye. It usually happens in the morning as I am awakening. It appears that I have sunglasses on, but just one eye? I have ask my eye specialist about this, but he doesn’t seem to have an answer.
I had a hard fall on my bottom yesterday 4 weeks after my catarac surgery i am 63 i am not seeing as well in that eye today and wondering if the fall is the reason ? i have no pain just blur & some double vision with reading i am not sure if it started before or after the fall my vision was 20/20 and clear until the last couple of days THANK YOU SO MUCH IHOPE TO HEAR FROM YOU! ROXY
Roxy wall, you need to see your cataract surgeon to look at your eye. There are many reasons … both from your surgery and neurologically … why you might have double vision. Call your eye surgeon.
i had cataract removed but no lens was placed by surgeon.
he said i had missing zonules.
what can i do
After 3 detached retinas in the last 10 months, I have an Aphakik eye. My surgeon is a truly Great Man, well respected and knowledgeable.
However I would like to know what the new implant is made from. The proposed new lens is from Alcon Labs. Acrylsof. I have read the information from the manufacturer and the clinical trails and it says safety is “reasonable” for three years. I have asked for a sample to get it analysed to see if any of the ingredients may cause me problems, but have come to a complete dead end. My surgeon just says he has had no problems in 15 years. Alcon wont even answer my mails.
If I buy a tin a baked beans then the ingredients are on the side! Am I being unreasonable in asking for the safety of an implant? Am I the only one? Any constructive comments gratefully received.
Paul,
The implants we use (I also use the Alcon lenses) are made of an acrylic plastic that has been in use for decades. I’ve never had a patient have a reaction to the material and have never heard of ANYONE having a reaction to this kind of plastic. Because of this, eye doctors don’t routinely discuss this possibility with their patients. There are so many OTHER things that could go wrong with a cataract surgery … the implant “material” is the aspect you should be LEAST worried about. With prior retinal detachment, you are at higher risk for lens dislocation, zonular dehiscence, re-detachment, capsular insufficiency, macular edema, corneal edema … you get the idea.
I searched around Alcon’s website, and found a PDF with some more data on the implants in question:
http://www.alconsurgical.com/pdfs/TOR240-DFU.pdf
Finally, I can see why your surgeon was hesitant to give you a “sample” … we don’t have “samples” to give out. These things are packaged in sterile boxes and somewhat expensive. The surgery center or hospital “owns” the hardware and if your doctor doesn’t own the center … he can’t walk over and grab one. Even then, he wouldn’t be able to get your medical insurance to cover the loss. Finally, I’m not sure what kind of testing you could even perform with such a small piece of plastic … it would fly off your finger if you breathed hard.
Good luck with your surgery!
Hi all. My son had a cataract surgery when he was 5 months. And At his 8 yrs old we decided to put a lenses to help him with vision and by not wearing thick eye glasses. But After Lense implant Doctor cleared many scar Tissue on my sons eye before implant was succesfully done. He had been prescribed with Prednisolone Acetate 1.0% and Ketorolac Tromethamine Opthalmic solution. And when they check the pressure of the eye it was high and prescribed with 2 new drops. and on the 2nd month to present he was still getting the 4 drops and pressure still didnt drop to normal eye pressure. I wanna know why my sons eye pressure didnt drop at all was the anti inflammatory drops are causing them not to make my sons eye pressure drop? and i read some articles that u cant use the 2 drops for more than a month. please give me idea on what should i know about. Thnks
4/8/14
Hello, hopefully someone can help!
Due to accident have been blind in left eye for 17 yrs. but 6 months ago a catatract developed in right eye and soon thereafter started loosing all vision bothclose and far.
Had Cataract Surgery 4/3/14 and right after vision was 99% both near & far next day Dr. said all lookks great and i was suprised i could see so well and was happy, i only had slight light sensetivity to headlights during day and all stop lights till i got up close.
Sat 3rd day after, got up and noticed stuff was fuzzy on walls and on TV i could no more make out small letters and faces were also messed up unless a close-upshot, then i drove and noticed that all road/street signs were un-readable unless i got right next to them, no matter how large they were.
Sunday am i called Dr and explained that in 2 days all went bad and my distance vision was the worst prob. since anything further than 4 feet was unfocused, i had a distance lens put in 21.0D but thought something was wrong,so she said come in Mon for checkup (1 week early).
Later Sunday i noticed with a lot of Blinking i could momentarily read letters in distance and the next morning on way to Dr. i could not make out any street signs unless i was totally at a stop and still could not read any above me at a light!
I had been told before surgery i would have only close-up vision probs and would need read classes, however for 2 days after Surgery, i needed nothingwhen i got to Dr. she tested my Vision and said my Close-up had gotten better than day after Surgery, but i explained on way down to her that all signs were fuzzy etc…..
She said it could take 6 weeks till i was healed from Surgery, but she did not explain why distance Vision had got so bad and seemed upset that i came in and told me to calm down.
I simply dont understand why with a lens for distance it had the opposite affect?
Could somebody explain if my probs are normal? Thank’s
John, when your surgeon chooses a lens to give you good “distance vision” there is no way to guarantee you’ll end up perfect. The lens calculations we use to pick your implant are based on average eyes, and everyone has different internal eye anatomy. Occasionally, someone will end up different than expected and still need glasses for both distance AND near vision.
Also, the implant tends to shift and settle into its final position in the eye over several weeks, so it is common for your focal point to shift during this time. This is why we don’t prescribe glasses for at least three weeks after cataract surgeon. Everything you have written sounds normal … just make sure you get your eye checked to rule out more serious problems like infection.
I just ended up watching all your video lectures. Currently studying for my specialty entrance exam (equivalent of the USMLE)
amazing work you have done. thank you very much. i wish you all the success and hope you make it as far as you wish and even more.
My daughter was born with a torn lens in her left eye. We discovered the blindness in that eye when she was 4 years old. After trying several treatments, we decided to remove the lens with hopes of getting vision through a contact lens. Unfortunately, we were not successful. In her “good eye” which is not so great, she has a “lazy, wondering eye”, and , now 15, she is more bothered by the appearance of this than the blind eye. So, my questions are: 1. We have been told differing opinions on the possibility of surgically replacing the lens to correct the blindness, what are your thoughts and what is the optimum age is surgery is possible? and, 2. Can her lazy eye be corrected by surgery and do you feel the risk might be too great? thanks so much!
My father (82) recently had cataract surgery. The doctor said he had a very small eye. Fluid was leaking at the incision site so they tried glue and it did not hold so they put in two stitches. Three weeks post op they remove one stitch and the fluid begins to leak again. He states it was to soon to remove the stitch. He then glued and placed a contact lens on the eye and states now the stitch that remains will be there for at least another month. Do the lens come in a size to fit your eye or are they all the same other than the strength of the lens. I am afraid this lens is to big and it will not seal at all. Any comments would be helpful.
Question- can lens replacement surgery be done a second time in the same eye?
After having eye with cataract/lens replacement vision is now only 20/40 and I still need long and reading sight of glasses.
Could I have second surgery to get the lens that doesn’t need long and reading sight glasses.
Plz sir suggest me
Thank you
when are you giong to upload its video ??
twenty some years ago I had cataract surgery. I am now having trouble with that eye. What chances are there that the lens is going bad?
Your article was very informative, thank you. Just one question: I have just had cataract surgery but the diopter is different from what was intended. Do the zonal fibers function when a new lens is inserted such that the diopter changes?
I had cataract surgery on both eyes several years ago. I was not given an option of having replacement lenses. Can lenses be instilled after cataract surgery?
Joe Wright – you don’t elaborate what you mean by “go bad”. See a specialist. Presumably you already know about capsule clouding, but if you don’t,that would be #1 possibility, fixed with trivial laser. Never mind the others, just see the specialist.
I love the book – and I am jealous of your many talents!!
On cataract surgery, for local anesthesia, you say “We can also perform a retrobulbar block by injecting lidocaine/bupivicane into the retrobulbar muscle cone”.
Is it true to say that the retrobulbar block procedure is no longer encouraged and now a subtenon block is much preferred as this have been proved to be safer?
You are to be commended for your talking your time to educate both students and the public. In today’s world, it is uncommon for a professional to publish an easily readable text on this normally esoteric subject.
Indeed, as I talked to a doctor who was interviewing me for cataract surgery, I had asked several questions which led him to get up and walk away, referring me to the young lady who was his assistant. He described the surgery several times as easy, even stating that I could within the first year remove the lens with no problem and change it out.
I realized if I was to feel comfortable, I’d have to start at the basics and gain a much better understanding of the eye composition as well as a more in-depth overview of the cataract procedure.
As you know, physicians generally treat their patients from an authoritarian viewpoint and my experience was quite negative. I started a technology company where we manufactured several types of hydrogels for use in various industries, having PhDs in polymer science working for me.
Although I am retired now, I can say I’ve never treated staff nor customers with a demeaning attitude. Frankly, it’s offending and speaks negatively of the persons character.
I find your writing quite helpful, and speaks quite well of your professionalism. Thanks!